| 1 |
GRONDAL C, JEANTY M, ENDERS D. Organocatalytic cascade reactions as a new tool in total synthesis[J].Nat Chem, 2010,2: 167-178.
|
| 2 |
GENC H N, OZGUN U, SIRI A. Design, synthesis and application of chiral tetraoxacalix[2] arene[2]triazine-based organocatalysts in asymmetric Michael addition reactions[J]. Chirality, 2019, 31(4): 1-8.
|
| 3 |
ATODIRESEI I, VILA C, RUEPING M. Asymmetric organocatalysis in continuous flow: opportunities for impacting industrial catalysis[J]. ACS Catal, 2015, 5(3): 1972-1985.
|
| 4 |
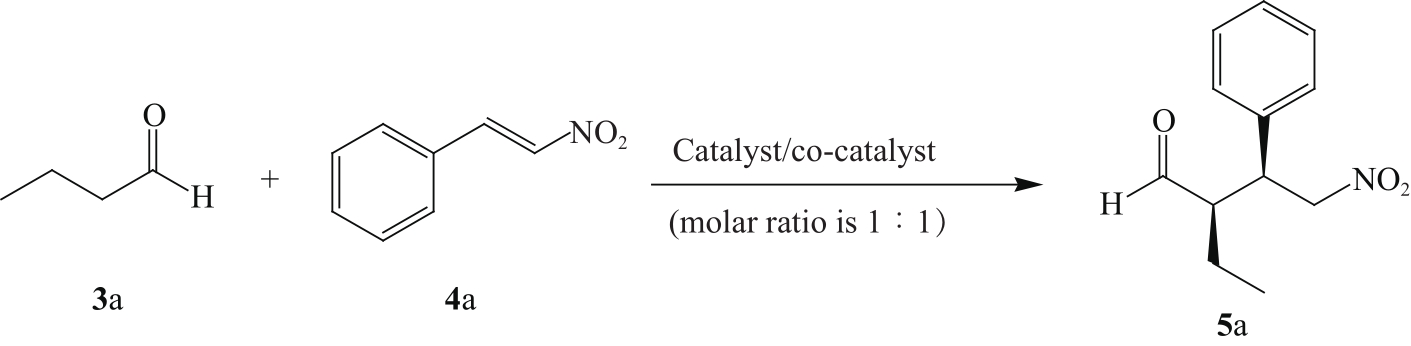
KUMAR T P, SATTAR M A, PRASAD S S, et al. Enantioselective Michael addition of aldehydes to nitroolefins catalyzed by pyrrolidine-HOBt[J]. Tetrahedron: Asymmetry, 2017, 28(3): 401-409.
|
| 5 |
ABOULENEIN M N, ELAZZOUNY A A, SALEH O A, et al. On chemical structures with potent antiepileptic/anticonvulsant profile[J]. Mini Rev Med Chem, 2012, 12(7): 671-700.
|
| 6 |
ANDRESEN H, AYDIN B E, MUELLER A, et al. An overview of gamma-hydroxybutyric acid: pharmacodynamics, pharmacokinetics, toxic effects, addiction, analytical methods, and interpretation of results[J]. Drug Test Anal, 2011, 3(9): 560-568.
|
| 7 |
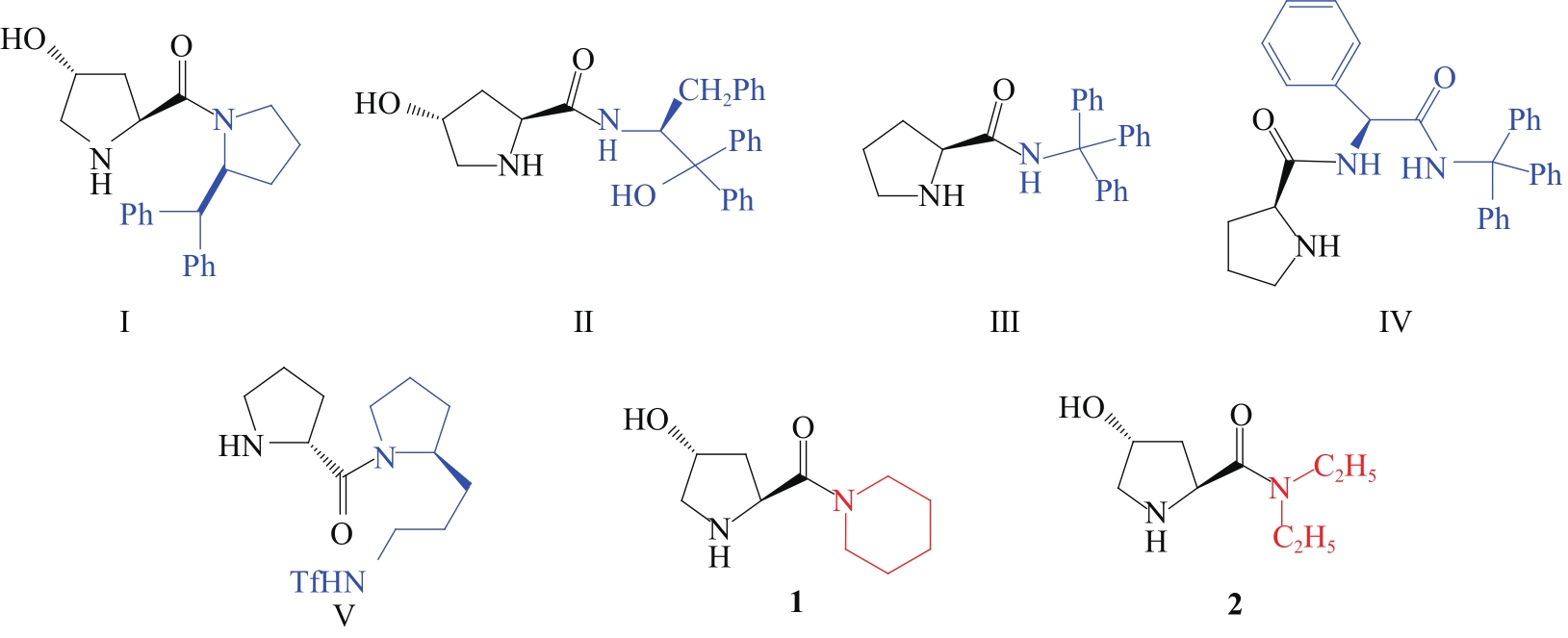
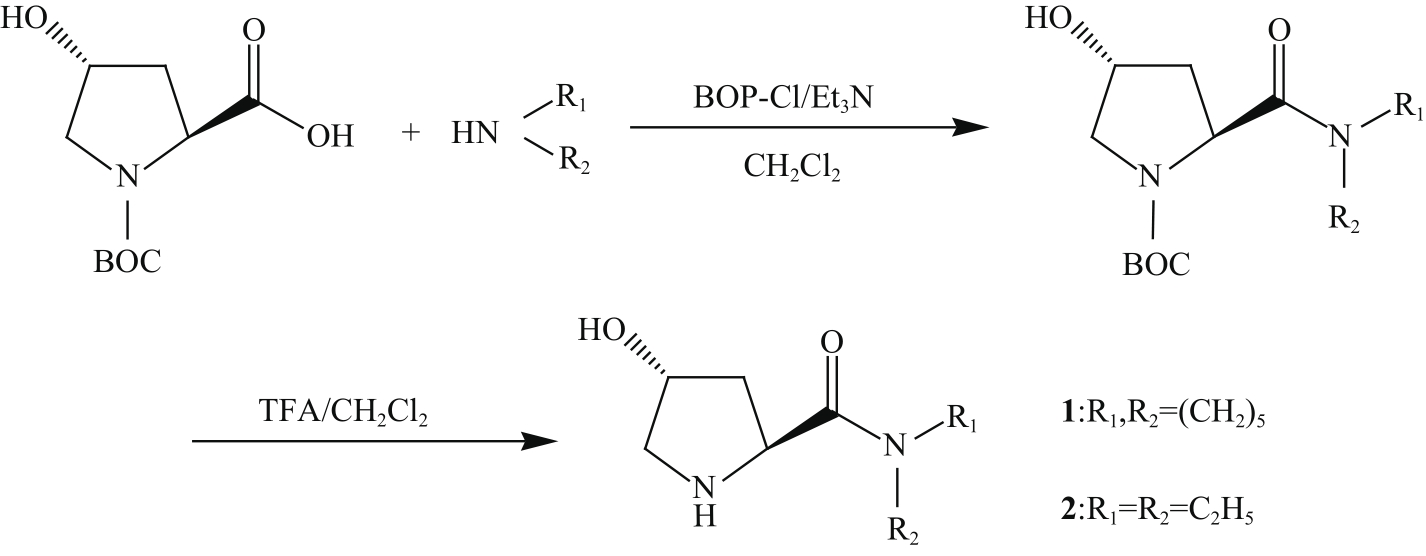
PALOMO C, VERA S, MIELGO A, et al. Highly efficient asymmetric Michael addition of aldehydes to nitroalkenes catalyzed by a simple trans-4-hydroxyprolylamide[J]. Angew Chem Int Ed, 2006, 45(36): 5984-5987.
|
| 8 |
OKUYAMA Y, NAKANO H, WATANABE Y, et al. Organocatalytic activity of 4-hydroxy-prolinamide alcohol with different noncovalent coordination sites in asymmetric Michael and direct aldol reactions[J]. Tetrahedron Lett, 2009, 50(2): 193-197.
|
| 9 |
WANG Y, LIN J, WEI K. Aromatic L-prolinamide-catalyzed asymmetric Michael addition of aldehydes to nitroalkenes[J]. Tetrahedron: Asymmetry, 2014, 25(24): 1599-1604.
|
| 10 |
XU D, WANG J, YAN L, et al. Novel bifunctional L-prolinamide derivatives as highly efficient organocatalysts for asymmetric nitro-Michael reactions[J]. Tetrahedron: Asymmetry, 2016, 27(22/23): 1121-1132.
|
| 11 |
GORDE A B, RAMAPANICKER R. D-Prolyl-2-(trifluoromethylsulfonamidopropyl)pyrrolidine: an organocatalyst for asymmetric Michael addition of aldehydes to β-nitroalkenes at ambient conditions[J]. J Org Chem, 2019, 84(3): 1523-1533.
|
| 12 |
YADAV G D, SINGH S. Prolinamide-catalysed asymmetric organic transformations[J]. Chem Select, 2019, 4(19): 5591-5618.
|
| 13 |
WANG Y, JI S, WEI K, et al. Epiandrosterone-derived prolinamide as an efficient asymmetric catalyst for Michael addition reactions of aldehydes to nitroalkenes[J]. RSC Adv, 2014, 4(58): 30850-30856.
|
| 14 |
WANG Y, LI D, LIN J, et al. Organocatalytic asymmetric Michael addition of aldehydes and ketones to nitroalkenes catalyzed by adamantoyl L-prolinamide[J]. RSC Adv, 2015, 5(8): 5863-5874.
|
| 15 |
NAZIROGLU H N, DURMAZ M, BOZKURT S, et al. Application of L-prolinamides as highly efficient organocatalysts for the asymmetric Michael addition of unmodified aldehydes to nitroalkenes[J]. Tetrahedron: Asymmetry, 2012, 23(2): 164-169.
|
| 16 |
LU D, GONG Y, WANG W. Prolylprolinol-catalyzed asymmetric Michael addition of aliphatic aldehydes to nitroalkenes[J]. Adv Synth Catal, 2010, 352(4): 644-650.
|
| 17 |
CHENG Y, BIAN Z, HE Y, et al. Asymmetric Michael addition of aldehydes to nitroolefins catalyzed by L-prolinamide derivatives using phenols as co-catalysts[J]. Tetrahedron: Asymmetry, 2009, 20(15): 1753-1758.
|
| 18 |
ZHU S, YU S, MA D. Highly efficient catalytic system for enantioselective Michael addition of aldehydes to nitroalkenes in water[J]. Angew Chem Int Ed, 2008, 47(3): 545-548.
|
| 19 |
LOMBARDO M, CHIARUCCI M, QUINTAVALLA A, et al. Highly efficient ion-tagged catalyst for the enantioselective Michael addition of aldehydes to nitroalkenes[J]. Adv Synth Catal, 2009, 351(17): 2801-2806.
|
| 20 |
ZHENG Z, PERKINS B L, NI B. Diarylprolinol silyl ether salts as new, efficient, water-soluble, and recyclable organocatalysts for the asymmetric Michael addition on water[J]. J Am Chem Soc, 2010, 132(1): 50-51.
|
| 21 |
WIESNER M, REVELL J F, WENNEMERS H. Tripeptides as efficient asymmetric catalysts for 1,4-addition reactions of aldehydes to nitroolefins-a rational approach[J]. Angew Chem Int Ed, 2008, 47(10): 1871-1874.
|
| 22 |
WIESNER M, UPERT G, ANGELICI G, et al. Enamine catalysis with low catalyst loadings-high efficiency via kinetic studies[J]. J Am Chem Soc, 2010, 132(1): 6-7.
|
| 23 |
WIESNER M, REVELL J D, TONAZZI S, et al. Peptide catalyzed asymmetric conjugate addition reactions of aldehydes to nitroethylene-a convenient entry into γ 2-amino acids[J]. J Am Chem Soc, 2008, 130(17): 5610-5611.
|
| 24 |
BORGES J, FEHER A, CRISLSTOMO F P, et al. Tetrahydropyran-based hybrid dipeptides as asymmetric catalysts for Michael addition of aldehydes to β-nitrostyrenes[J]. Adv Synth Catal, 2017, 359(4): 576-583.
|
| 25 |
BORGES J, GARCIA I, MARTIN T. Conformational control of tetrahydropyran-based hybrid dipeptide catalysts improves activity and stereoselectivity[J]. Adv Synth Catal, 2019, 361(9): 2141-2147.
|
| 26 |
BIEL M, DECK P, GIANNIS A, et al. Synthesis and evaluation of acyl protein thioesterase 1(APT1)inhibitors[J]. Chem Eur J, 2006, 12(15): 4121-4143.
|
| 27 |
BETANCORT J M, BARBAS C F. Catalytic direct asymmetric michael reactions: taming naked aldehyde donors[J]. Org Lett, 2001, 3(23): 3737-3740.
|

 ), Zheng BIAN2(
), Zheng BIAN2( )
)