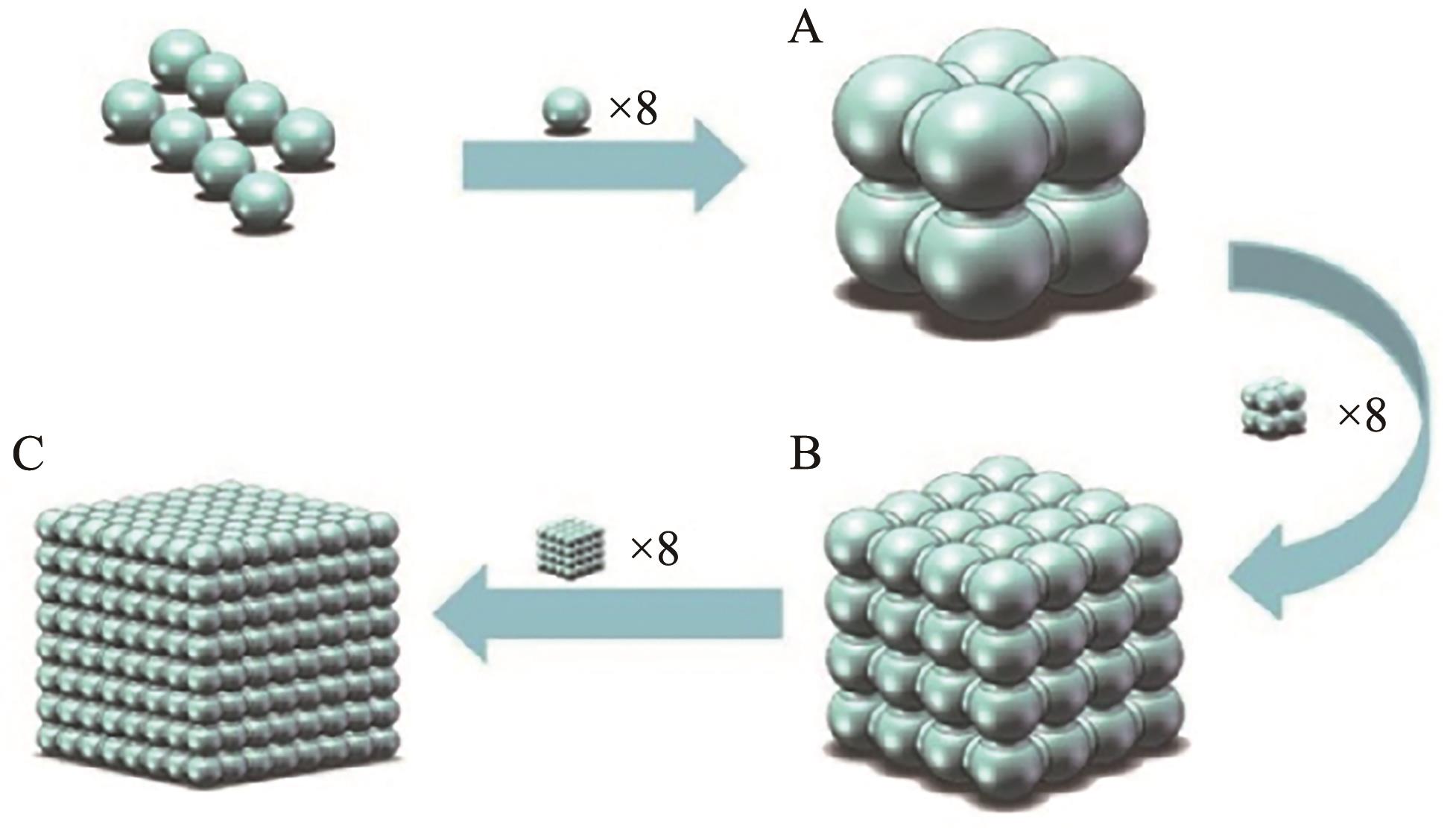
Chinese Journal of Applied Chemistry ›› 2023, Vol. 40 ›› Issue (12): 1726-1736.DOI: 10.19894/j.issn.1000-0518.230151
• Full Papers • Previous Articles Next Articles
Simulation of Aggregate Structure Evolution of Calcium-Based Absorbent Based on Sintering Mechanism
Jie JI, Ming-Chun LI( ), Cun-Liang SHEN, Xin YANG
), Cun-Liang SHEN, Xin YANG
- College of Materials Science and Engineering,Shenyang University of Technology,Shenyang 110023,China
-
Received:2023-05-22Accepted:2023-11-13Published:2023-12-01Online:2024-01-03 -
Contact:Ming-Chun LI -
About author:mingchunlihlj@163.com
-
Supported by:the National Natural Science Foundation of China(51874200);Liaoning Bai Qian Wan Talents Program(XLYC2008014);Liaoning Revitalization Talents Program(XLYC1907080)
CLC Number:
Cite this article
Jie JI, Ming-Chun LI, Cun-Liang SHEN, Xin YANG. Simulation of Aggregate Structure Evolution of Calcium-Based Absorbent Based on Sintering Mechanism[J]. Chinese Journal of Applied Chemistry, 2023, 40(12): 1726-1736.
share this article
Add to citation manager EndNote|Ris|BibTeX
URL: http://yyhx.ciac.jl.cn/EN/10.19894/j.issn.1000-0518.230151
| Parameter | Ds/(cm2·s-1)[ | Dv/(cm2·s-1)[ | Ω/cm3 [ | ψ/(°) | k/(J·K-1) | |
|---|---|---|---|---|---|---|
| Value | 8.5×10-11 | 1.667×10-15 | 1.5×10-4 | 1.72×10-23 | 5π/6~π | 1.38×10-23 |
Table 1 Calculation parameters in sintering model
| Parameter | Ds/(cm2·s-1)[ | Dv/(cm2·s-1)[ | Ω/cm3 [ | ψ/(°) | k/(J·K-1) | |
|---|---|---|---|---|---|---|
| Value | 8.5×10-11 | 1.667×10-15 | 1.5×10-4 | 1.72×10-23 | 5π/6~π | 1.38×10-23 |
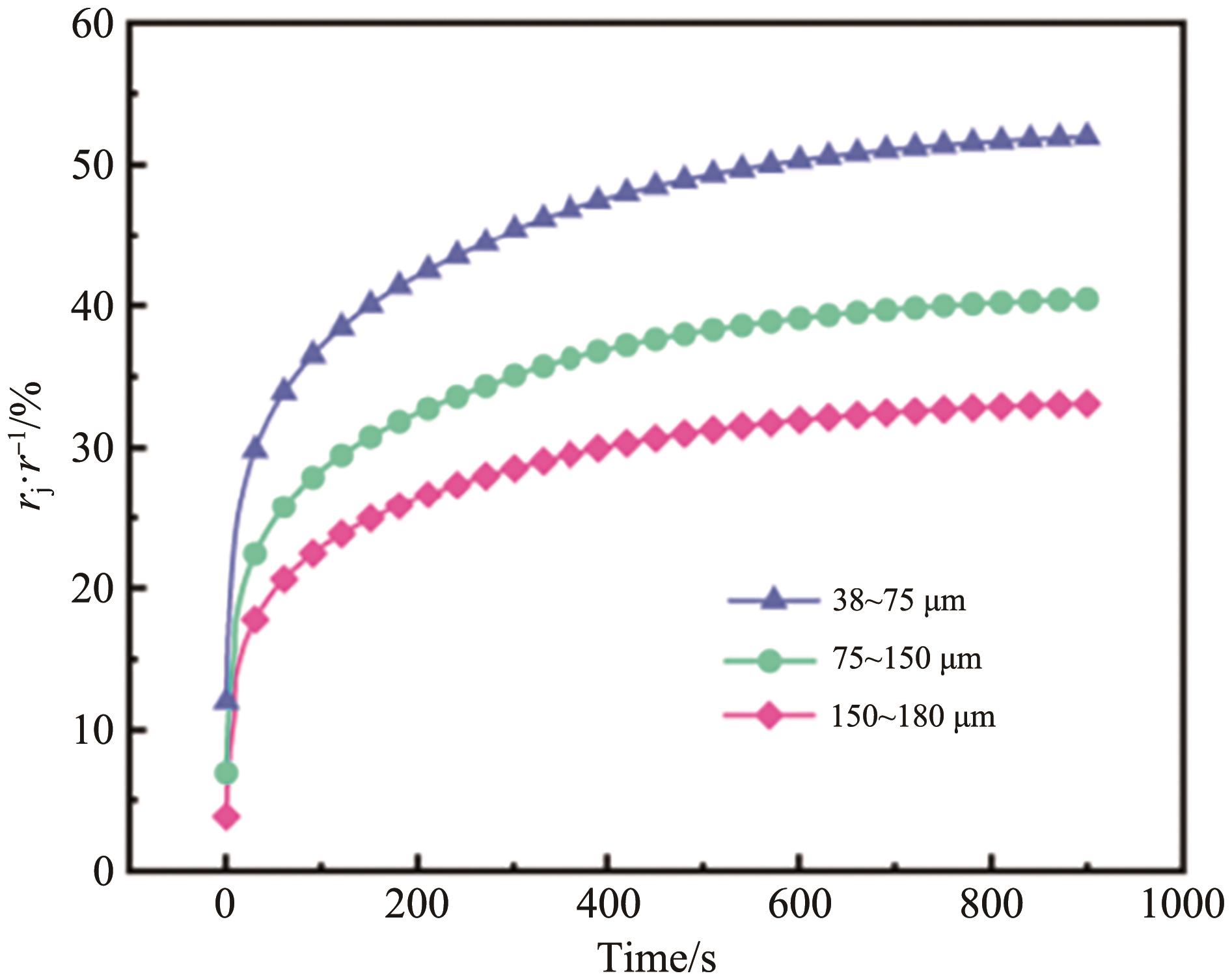
| Time/s | rj·r-1 (38~75 μm)/% | rj·r-1 (75~150 μm)/% | rj·r-1 (150~180 μm)/% |
|---|---|---|---|
60 300 600 | 33.81 45.24 50.30 | 25.66 34.98 39.09 | 20.55 28.55 31.93 |
Table 2 Sintering neck growth rate of different particle size sorbents at different time periods
| Time/s | rj·r-1 (38~75 μm)/% | rj·r-1 (75~150 μm)/% | rj·r-1 (150~180 μm)/% |
|---|---|---|---|
60 300 600 | 33.81 45.24 50.30 | 25.66 34.98 39.09 | 20.55 28.55 31.93 |
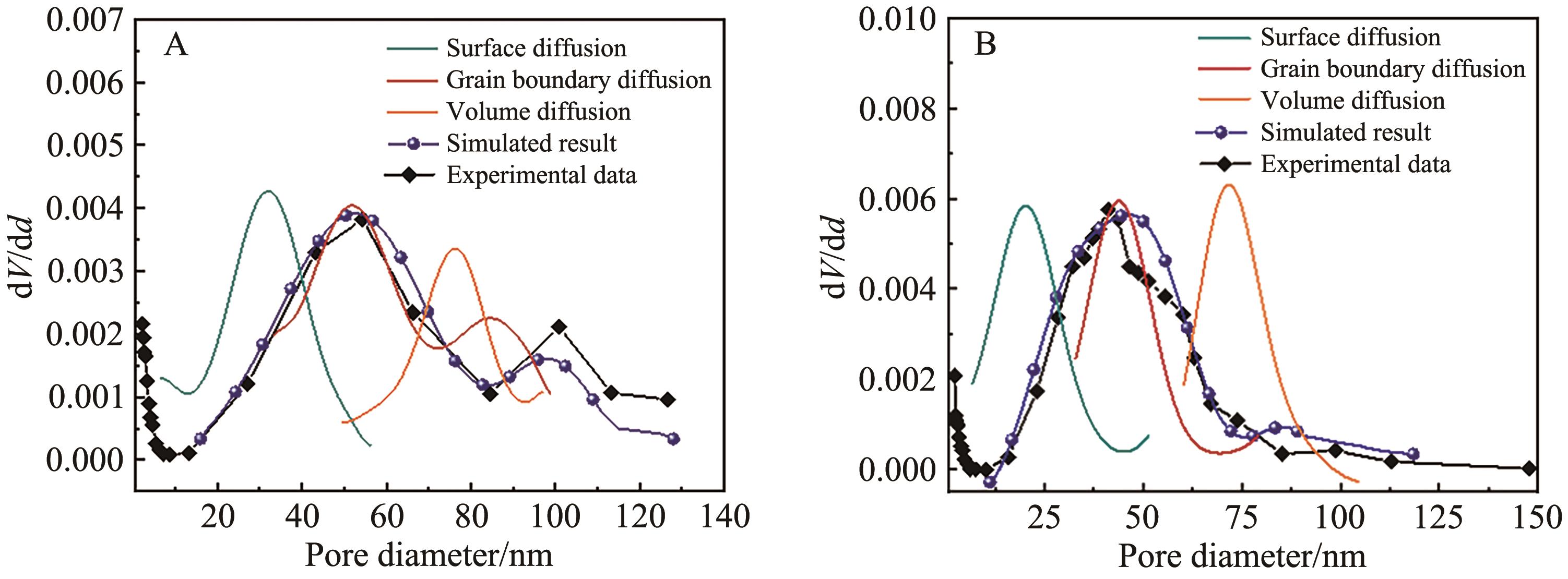
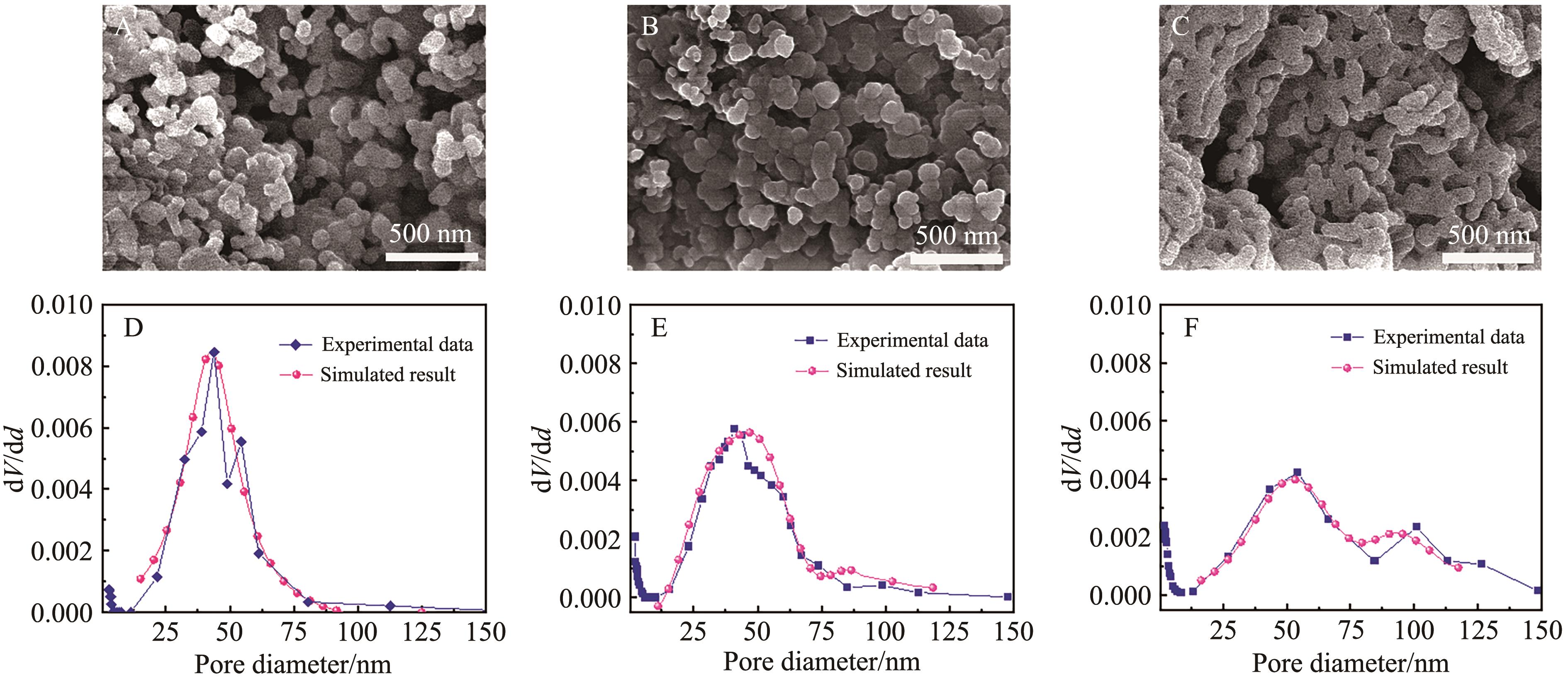
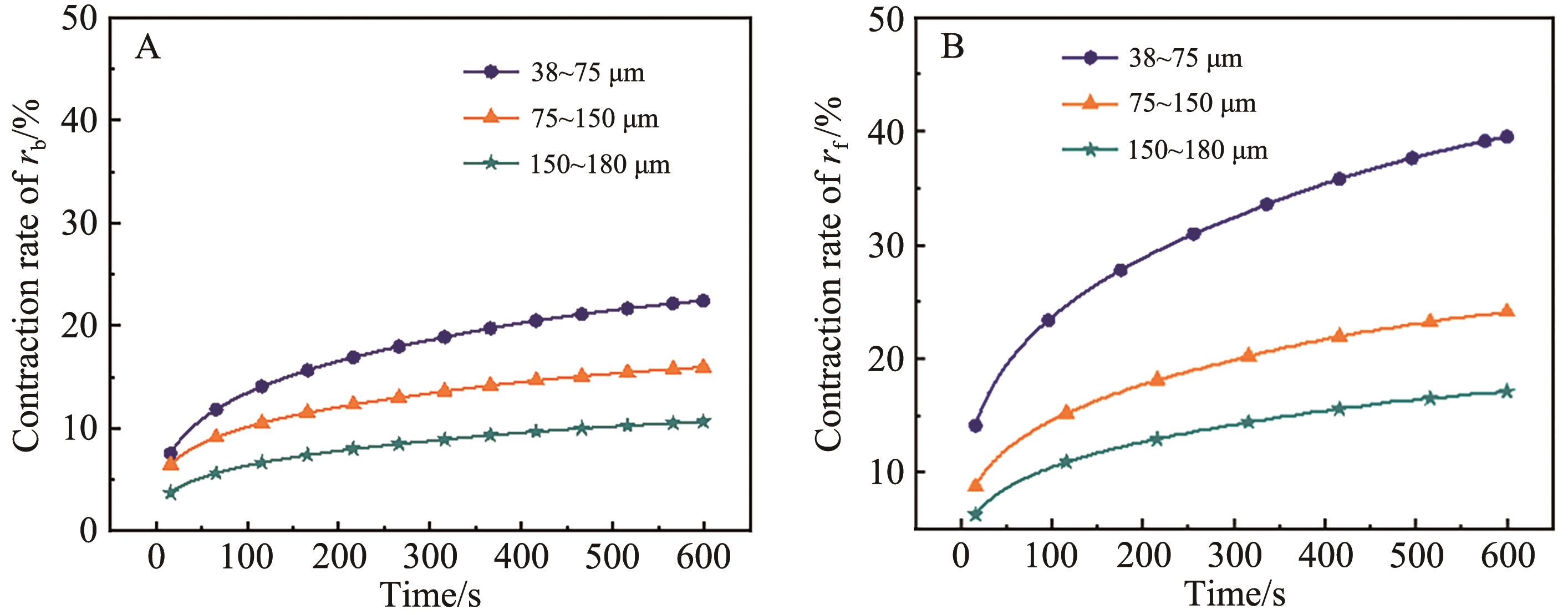
Fig.6 Model calculation results and experimental measurement results of structural evolution of sintered for 300 s pore under the control of different sintering mechanisms of calcium absorbers with different particle sizes of (A) 38~75 μm and (B) 75~150 μm

Fig.7 Model calculation results and experimental measurement results of structural evolution of sintered 600 s pore under the control of different sintering mechanisms of calcium absorbers with different particle sizes of (A) 38~75 μm and (B) 75~150 μm
| 1 | JING J, LI T, ZHANG X, et al. Enhanced CO2 sorption performance of CaO/Ca3Al2O6 sorbents and its sintering-resistance mechanism[J]. Appl Energy, 2017, 199: 225-233. |
| 2 | CHENG L, DUAN, L. From waste to high value utilization of spent bleaching clay in synthesizing I high-performance calcium-based sorbent for CO2 capture[J] Appl Energy, 2018, 210: 117-126. |
| 3 | BHATIA K, PERLMUTTER D. Effect of the product layer on the kinetics of the CO2-lime reaction[J]. Aiche J, 2010, 29(1): 79-86. |
| 4 | ALVAREZ D, ABANADES C. Determination of the critical product layer thickness in the reaction of CaO with CO2[J]. Ind Eng Chem Res, 2005, 44(15): 5608-5615. |
| 5 | VALVERDE M, RAGANATI F, QUINTANILLA A, et al. Enhancement of CO2 capture at Ca-looping conditions by high-intensity acoustic fields[J]. Appl Energy, 2016, 111(2): 538-549. |
| 6 | KUCZYNSKI C. Self-diffusion in sintering of metallic particles[J]. Jom, 1949, 1(2): 169-178. |
| 7 | GERMAN M. Surface area reduction kinetics during intermediate stage sintering[J]. J Am Cerm Soc, 2010, 61(5/6): 272-274. |
| 8 | COBLE L. Sintering crystalline solids: I. intermediate and final stage models; II. experimental test of diffusion models in powder compacts[J]. J Phys D Appl Phys, 1961, 32(5): 787-792. |
| 9 | 施剑林. 固相烧结—Ⅱ粗化与致密化关系及物质传输途径[J]. 硅酸盐学报, 1997, 25(6): 657-668. |
| SHI J L. Solid phase sintering—Ⅱ coarsening and densification relationship and material transport pathway[J]. J Chin Ceram Soc, 1997, 25(6): 657-668. | |
| 10 | BLAMEY J, SHI J, ZHONG X, et al. A novel calcium looping absorbent incorporated with polymorphic spacers for hydrogen production and CO2 capture[J]. Energy Environ Sci, 2014, 7(10): 3291-3295. |
| 11 | ABANADES C, ALVAREZ D. Conversion limits in the reaction of CO2 with lime[J]. Energy Fuels, 2003, 17(2): 308-315. |
| 12 | CHEN H, ZHAO Z, HUANG X, et al. Novel optimized process for utilization of CaO-based sorbent for capturing CO2 and SO2 sequentially[J]. Energy Fuels, 2012, 26(9/10): 5596-5603. |
| 13 | GULLRTT K, BRUCE R. Pore distribution changes of calcium-based sorbents reacting with sulfur dioxide[J]. Aiche J, 2010, 33(10):1719-1726. |
| 14 | 乔春珍, 王宝利, 肖云汉. 钙基CO2吸收剂循环活性衰减原因初探[J]. 化工学报, 2010, 61(3): 720-724. |
| QIAO C Z, WANG B L, XIAO Y H. Preliminary study on the reasons for the decline of circulating activity of calcium based CO2 absorbent[J]. J Chem Eng, 2010, 61(3): 720-724. | |
| 15 | IYER V, GUPTA H, SAKADJIAN B, et al. Multicyclic study on the simultaneous carbonation and sulfation of high-reactivity CaO[J]. Ind Eng Chem Res, 2002, 43(14): 3939-3947. |
| 16 | SUN P, GRACE R, LIM C, et al. The effect of CaO sintering on cyclic CO2 capture in energy systems[J]. Aiche J, 2007, 53(9): 2432-2442. |
| 17 | ALVAREZ D, ABANADES C. Determination of the critical product layer thickness in the reaction of CaO with CO2[J]. Ind Eng Chem Res, 2005, 44(15): 5608-5615. |
| 18 | FENNELL S, PACCIANI R, DENNIS S, et al. The effects of repeated cycles of calcination and carbonation on a variety of different limestones, as measured in a hot fuidized bed of sand[J]. Energy Fuels, 2007, 21(4): 2072-2081. |
| 19 | 王春波, 周兴, 张斌, 等. 烧结对钙基吸收剂循环煅烧/碳酸化捕集CO2影响[J]. 中国电机工程学报, 2014, 34(35): 8. |
| WANG C B, ZHOU X, ZHANG B, et al. Effect of sintering on CO2 capture by calcination/carbonation with calcium absorbent[J]. Pro CSEE, 2014, 34(35): 8. | |
| 20 | 郑之民, 周兴, 王春波. O2/CO2气氛下钙基脱硫剂孔隙分布模拟[J]. 电力科学与工程, 2013, 29(5): 67-70. |
| ZHENG Z M, ZHOU X, WANG C B. Simulation of pore distribution of calcium based desulfurizer in O2/CO2 atmosphere[J]. Electric Power Sci Eng, 2013, 29(5): 67-70. | |
| 21 | SILCOX D. Amathematical model for the flash calcinations of dispersed CaCO3 and Ca(OH)2 particles[J]. Ind Eng Chem Res, 1989(28): 155-160. |
| 22 | GHOSH DASTIDAR A, MAHLI S, AGNIHOTRI R, et al. Ultrafast calcination and sintering of Ca(OH)2 powder: experimental and modeling[J]. Chem Int J Eng Sci, 1995, 50(13): 2029-2040. |
| 23 | BHATTACHARYA A, PUROHIT P. Predicting reaction rates for non-catalytic fluid-solid reactions in presence of structural changes in the solid phase[J]. Chem Eng J, 2004, 102(2): 141-149. |
| 24 | MAYA C, JANNA C. Novel model for non-catalytic solid-gas reactions with structural changes by chemical reaction and sintering[J]. Chem Eng Sci, 2016, 142: 258-268. |
| 25 | LI Z, OU J, LUO G, et al. High-efficiency CaO-based sorbent modified by aluminate cement and organic fiber through wet mixing method[J]. Ind Eng Chem Res, 2019, 58(48): 22040-22047. |
| 26 | YOUNG T. Miscellaneous works.vol 1, metal powder industries federation[M]. New Jersey: Princeton, 1980: 58-60. |
| 27 | 果世驹. 粉末冶金原理[M]. 北京: 冶金工业出版社, 1997: 272-283. |
| GUO S J. Principle of powder metallurgy[M]. Beijing: Metallurgical Industry Press, 1997: 272-283. | |
| 28 | WAKAI F, ALDINGER F. Sintering through surface motion by the difference in mean curvature[J]. Acta Mater, 2003, 51(14): 4013-4024. |
| 29 | WAKAI F, ALDINGER F. Sintering forces in equilibrium and non-equilibrium states during sintering of two particles[J]. Sci Technol Adv Mat, 2004, 5(4): 521-525. |
| 30 | PAN J, COCKS C. A numerical technique for the analysis of coupled surface and grain-boundary diffusion[J]. Acta Metall Et Mater, 1995, 43(4): 1395-1406. |
| 31 | KUCHCHERENKO S, PAN J, YEOMANS A. A combined finite element and finite difference scheme for computer simulation of microstructure evolution and its application to pore-boundary separation during sintering[J]. Comput Mater Sci, 2000, 18(1): 76-92. |
| 32 | GOUVEA D, CASTRO R. Sintering: the role of interface energies[J]. Appl Surf Sci, 2003, 217(1/4): 194-201. |
| 33 | SVOBODA J, RIEDEL H. New solutions describing the formation of interparticle necks in solid-state sintering[J]. Acta Metall Et Mater, 1995, 43(1): 1-10. |
| 34 | SRINIVASAN R, TRIVEDI R. Theory of grain boundary grooving under the combined action of the surface and volume diffusion mechanisms[J]. Acta Metall Sin Engl, 1973, 21(5): 611-620. |
| 35 | HORIYA T, ISHIKAWA M, YAMAJI K, et al. Calcium tracer diffusion in (La,Ca)CrO3 by SIMS[J]. Solid State Ionics, 1999,124(3/4): 301-307. |
| 36 | 左骁遥, 房晓红, 曾凡桂. 二氧化碳在高岭石孔隙中吸附的分子模拟[J]. 矿产综合利用, 2020, 1(5): 36. |
| ZUO X Y, FANG X H, ZENG F G. Molecular simulation of carbon dioxide adsorption in kaolinite pores[J]. Compr Util Min, 2020, 1(5): 36. |
| No related articles found! |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||