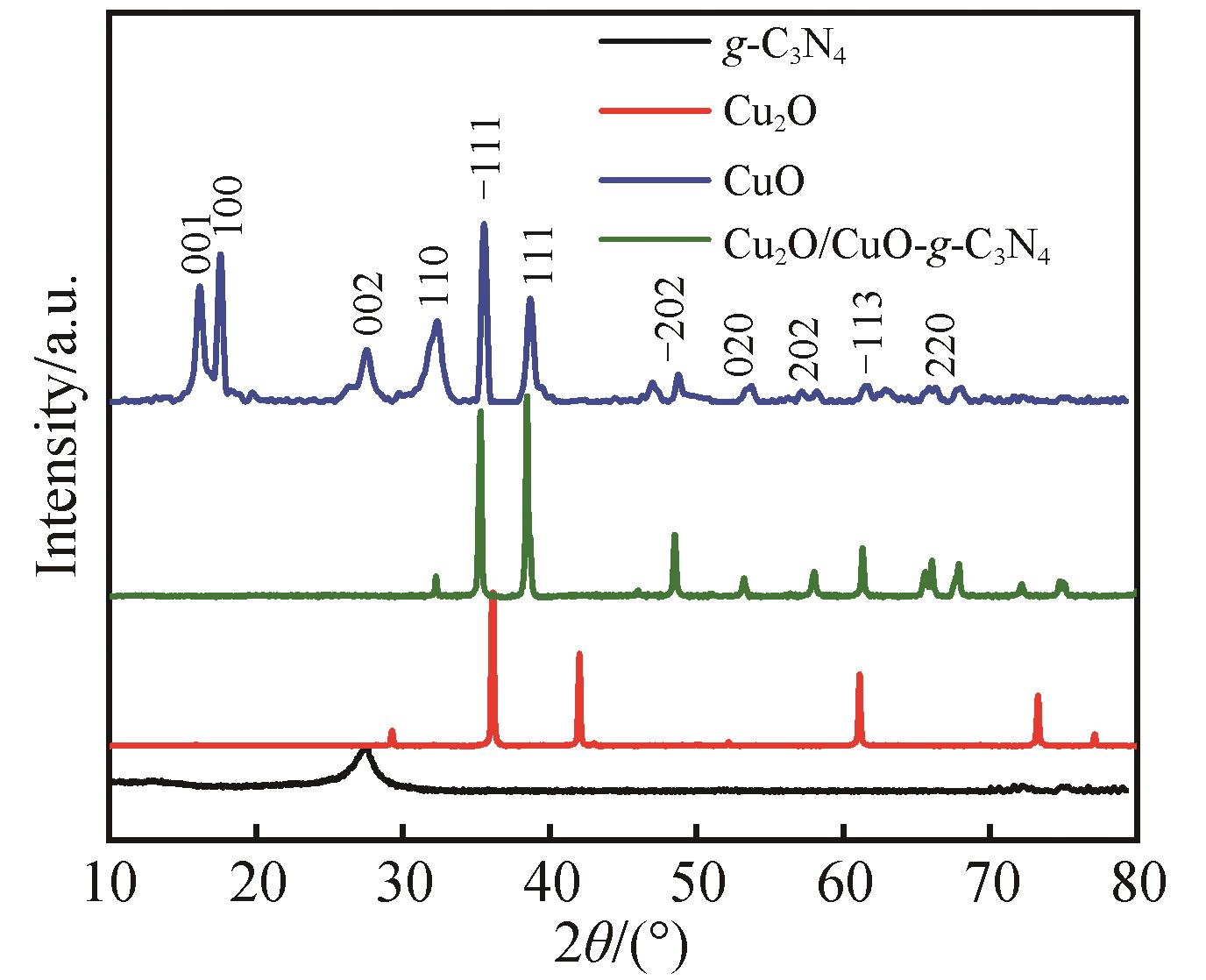
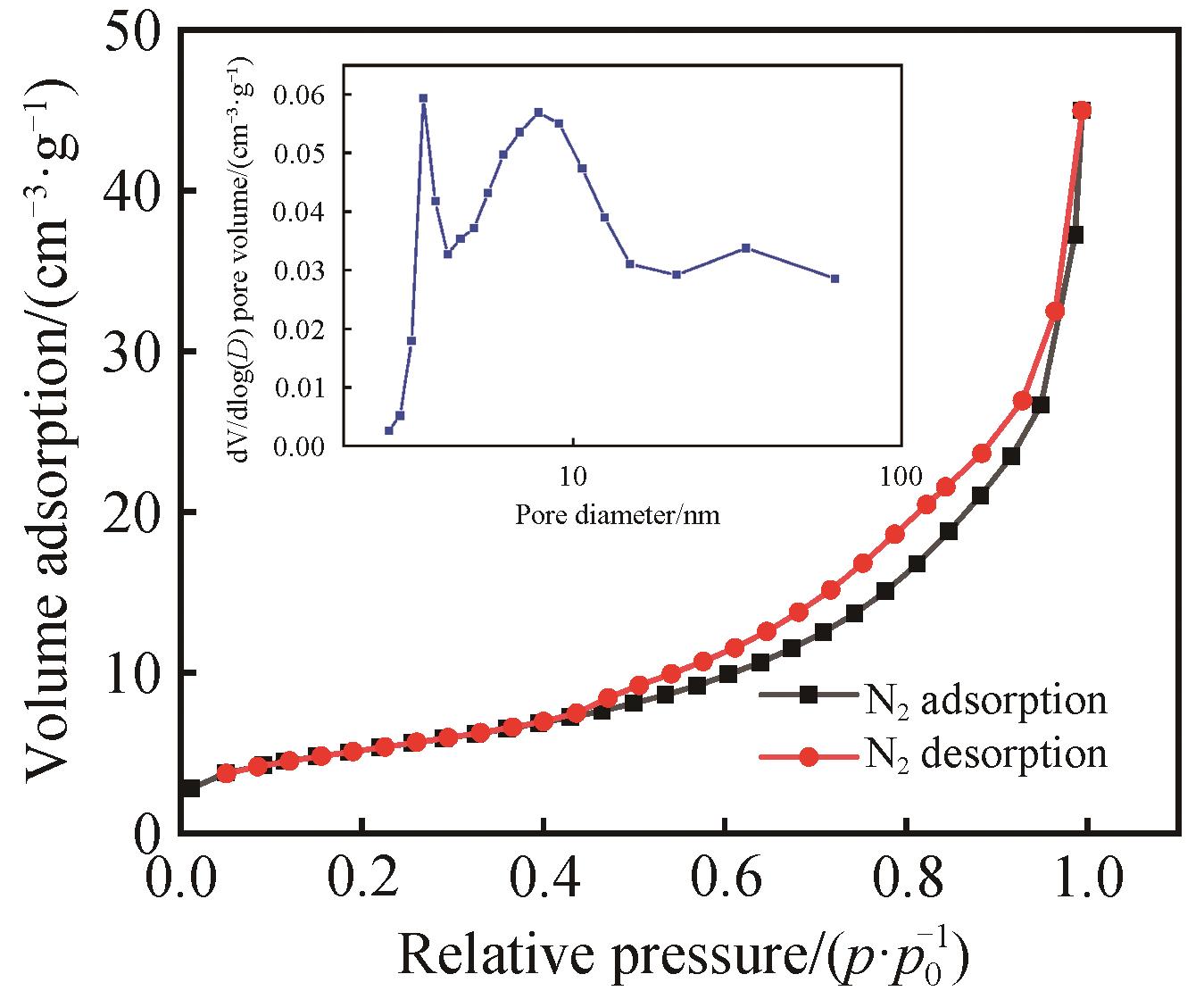
Chinese Journal of Applied Chemistry ›› 2023, Vol. 40 ›› Issue (3): 420-429.DOI: 10.19894/j.issn.1000-0518.220235
• Full Papers • Previous Articles Next Articles
Preparation of Cu2O/CuO-g-C3N4 Adsorbent by One-step Thermal Polymerization and Adsorption Properties for Methyl Orange
Bo XIONG, Tai-Hua LI, Wu-Ping ZHOU, Chang-Yu LIU( ), Xiao-Long XU(
), Xiao-Long XU( )
)
- School of Biotechnology and Health Sciences,Wuyi University,Jiangmen 529020,China
-
Received:2022-07-08Accepted:2023-02-19Published:2023-03-01Online:2023-03-27 -
Contact:Chang-Yu LIU,Xiao-Long XU -
About author:xyxl@wyu.edu.cn
wyuchemcyliu@126.com;
-
Supported by:the National Natural Science Foundation of China(21974097);the Education Department of Guangdong Province(2020KSYS004);Guangdong Province's Special Fund for Science and Technology Innovation Strategy (College Students' Science and Technology Innovation Cultivation)(pdjh2022b0531);the Science and Technology Bureau of Jiangmen(2019030102360012639);the Undergraduate Training Programs of Wuyi University(202111349019)
CLC Number:
Cite this article
Bo XIONG, Tai-Hua LI, Wu-Ping ZHOU, Chang-Yu LIU, Xiao-Long XU. Preparation of Cu2O/CuO-g-C3N4 Adsorbent by One-step Thermal Polymerization and Adsorption Properties for Methyl Orange[J]. Chinese Journal of Applied Chemistry, 2023, 40(3): 420-429.
share this article
Add to citation manager EndNote|Ris|BibTeX
URL: http://yyhx.ciac.jl.cn/EN/10.19894/j.issn.1000-0518.220235
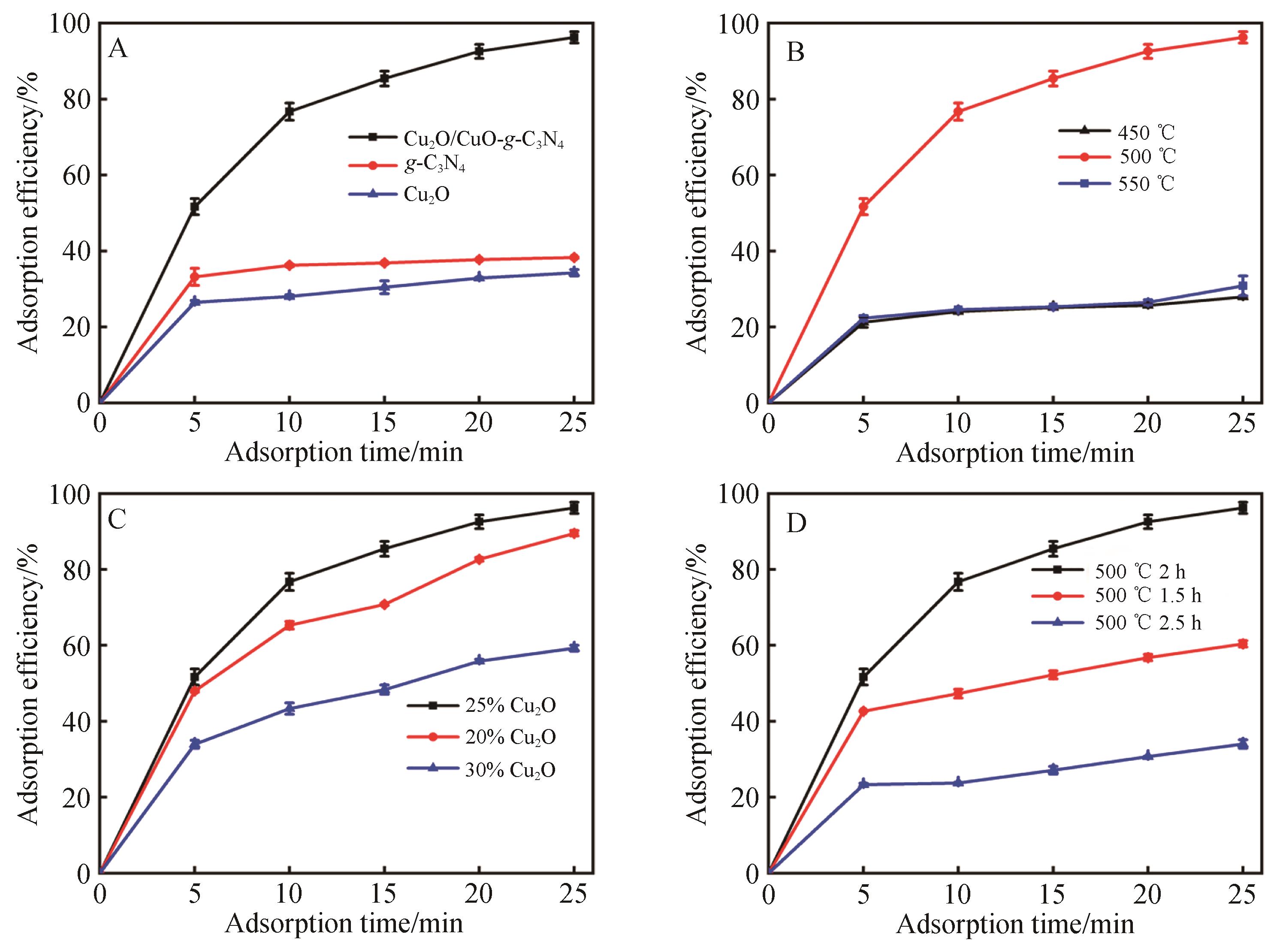
Fig.5 Adsorption efficiency for different (A) adsorbents, (B) polymerization temperature, (C) Cu2O dosage(w% is mass fraction of Cu2O in precursor) and (D) polymerization duration at 500 ℃ (20 mg adsorbent added into 100 mL 30 mg/L MO solution, pH = 6.0, 1400 r/min)
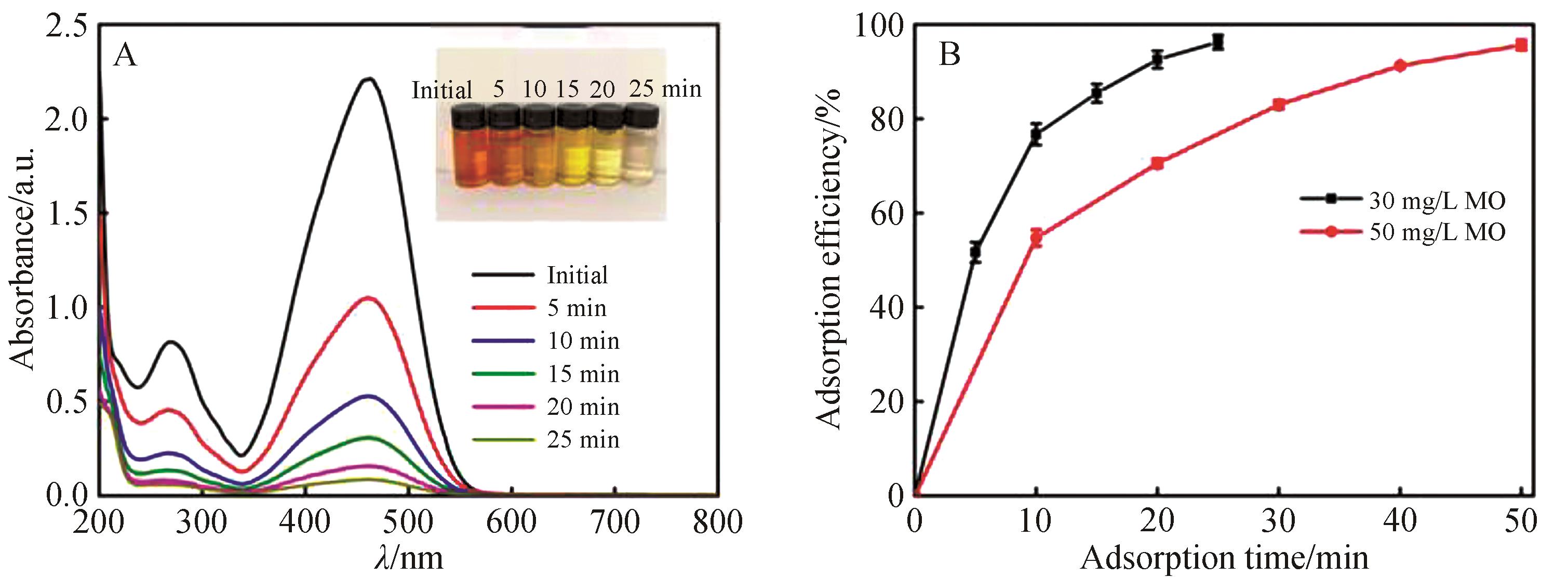
Fig.6 UV-Vis spectra (A) and photos (inset) of 30 mg/L MO solution with different adsorption duration; (B) adsorption efficiency with different MO concentration (20 mg adsorbent added into 100 mL MO solution, pH=6.0, 1400 r/min)
| Graphene-based nanomaterial | Qe/(mg·g-1) | Ref. |
|---|---|---|
| PmPD/rGO/Nickle ferrite | 136 | [ |
| Al-doped CNTs | 69.70 | [ |
| Cu-Mo MOFs | 48.13 | [ |
| Clay | 41.67 | [ |
| Nitrogen-doped graphene gels | 232 | [ |
| rGO | 224 | [ |
| Cu2O/CuO-g-C3N4 | 241.25 | This work |
Table 1 Equilibrium adsorption amount comparation between this work and references
| Graphene-based nanomaterial | Qe/(mg·g-1) | Ref. |
|---|---|---|
| PmPD/rGO/Nickle ferrite | 136 | [ |
| Al-doped CNTs | 69.70 | [ |
| Cu-Mo MOFs | 48.13 | [ |
| Clay | 41.67 | [ |
| Nitrogen-doped graphene gels | 232 | [ |
| rGO | 224 | [ |
| Cu2O/CuO-g-C3N4 | 241.25 | This work |
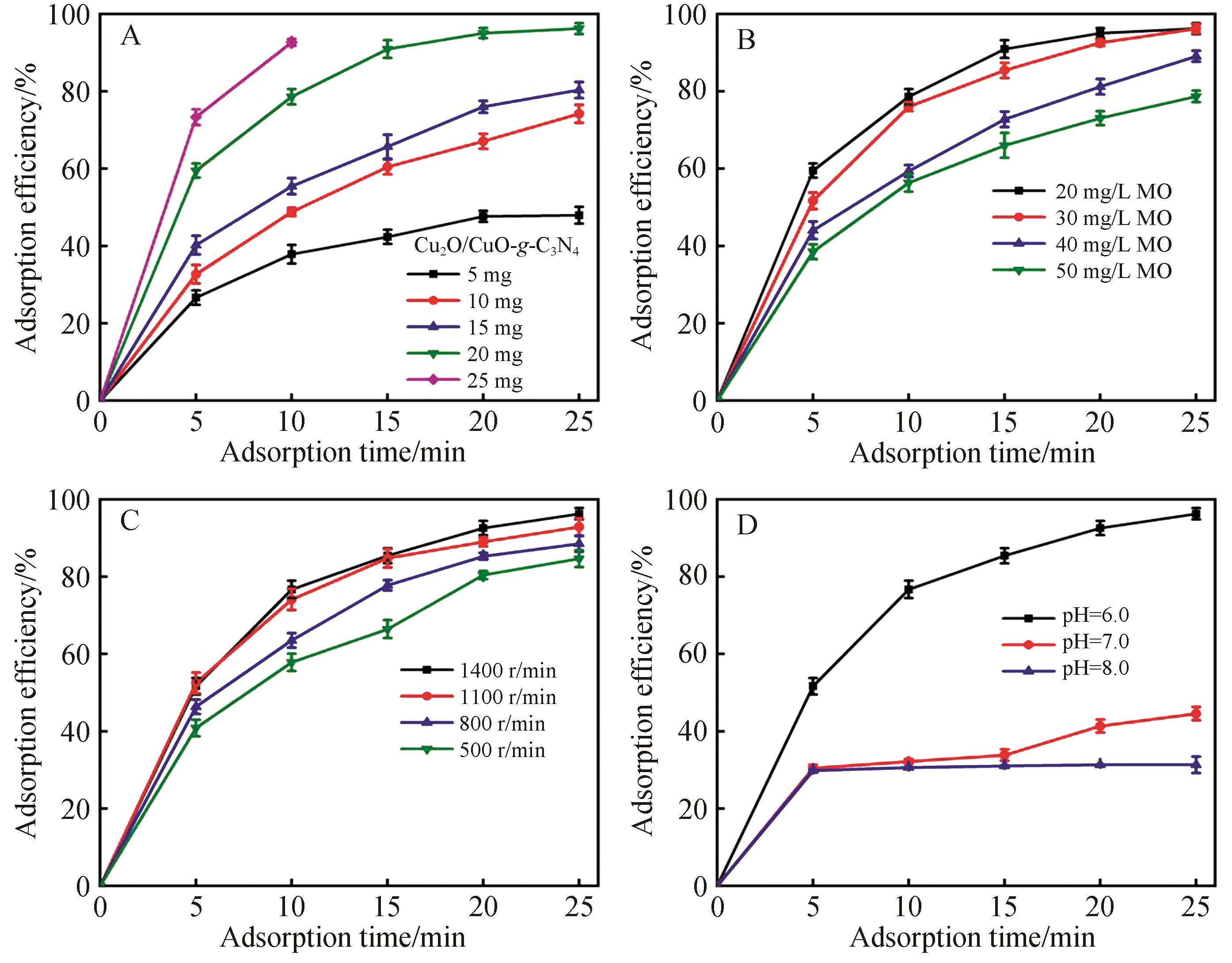
Fig.7 Adsorption efficiency with different (A) dosage of Cu2O/CuO-g-C3N4, (B) MO concentration, (C) stirring rate and (D) pH (All experiments are executed at 20 mg adsorbent add into 100 mL 30 mg/L MO solution, pH=6.0 and stirring velocity of 1400 r/min unless mentioned otherwise)
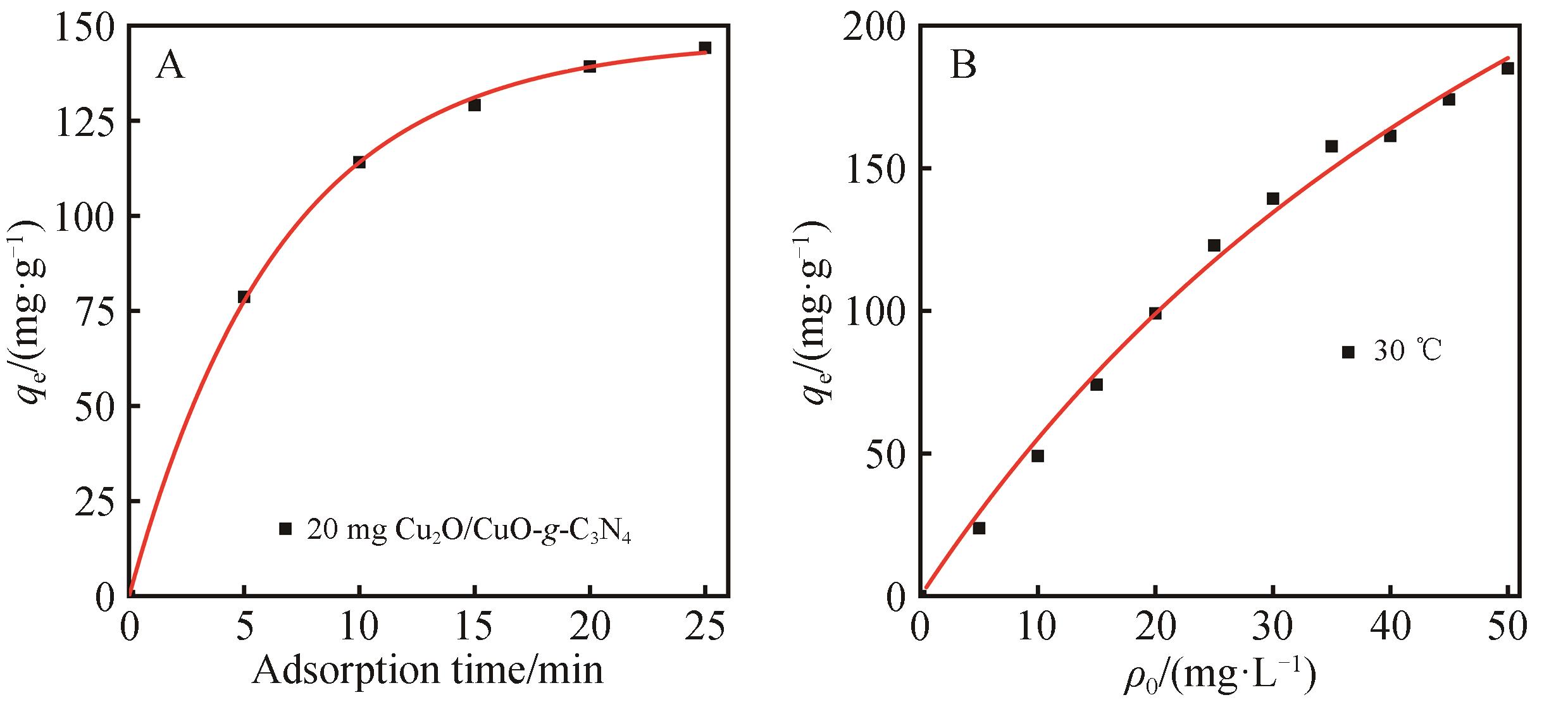
Fig.8 Adsorption kinetics (A) and isotherm curve (B) of MO on the Cu2O/CuO-g-C3N4 absorbent ((A) 20 mg adsorbent added into 100 mL 30 mg/L MO solution, pH=6.0, 1400 r/min and (B) 20 mg adsorbent added into 100 mL MO solution, pH=6.0, 180 r/min shaking)
| Preudo-first-order model | ||||
|---|---|---|---|---|
| ρ0/(mg·L-1) | K1 | qe/(mg·g-1) | r | |
| MO | 30 | 0.004 2 | 144.75 | 0.999 |
Table 2 Kinetic parameter fitting of MO adsorption
| Preudo-first-order model | ||||
|---|---|---|---|---|
| ρ0/(mg·L-1) | K1 | qe/(mg·g-1) | r | |
| MO | 30 | 0.004 2 | 144.75 | 0.999 |
| Langmuir constants | ||
|---|---|---|
| qe/(mg·g-1) | KL/(L·mg-1) | r |
| 185 | 0.002 3 | 0.990 |
Table 3 Parameters for Langmuir model for MO adsorption
| Langmuir constants | ||
|---|---|---|
| qe/(mg·g-1) | KL/(L·mg-1) | r |
| 185 | 0.002 3 | 0.990 |
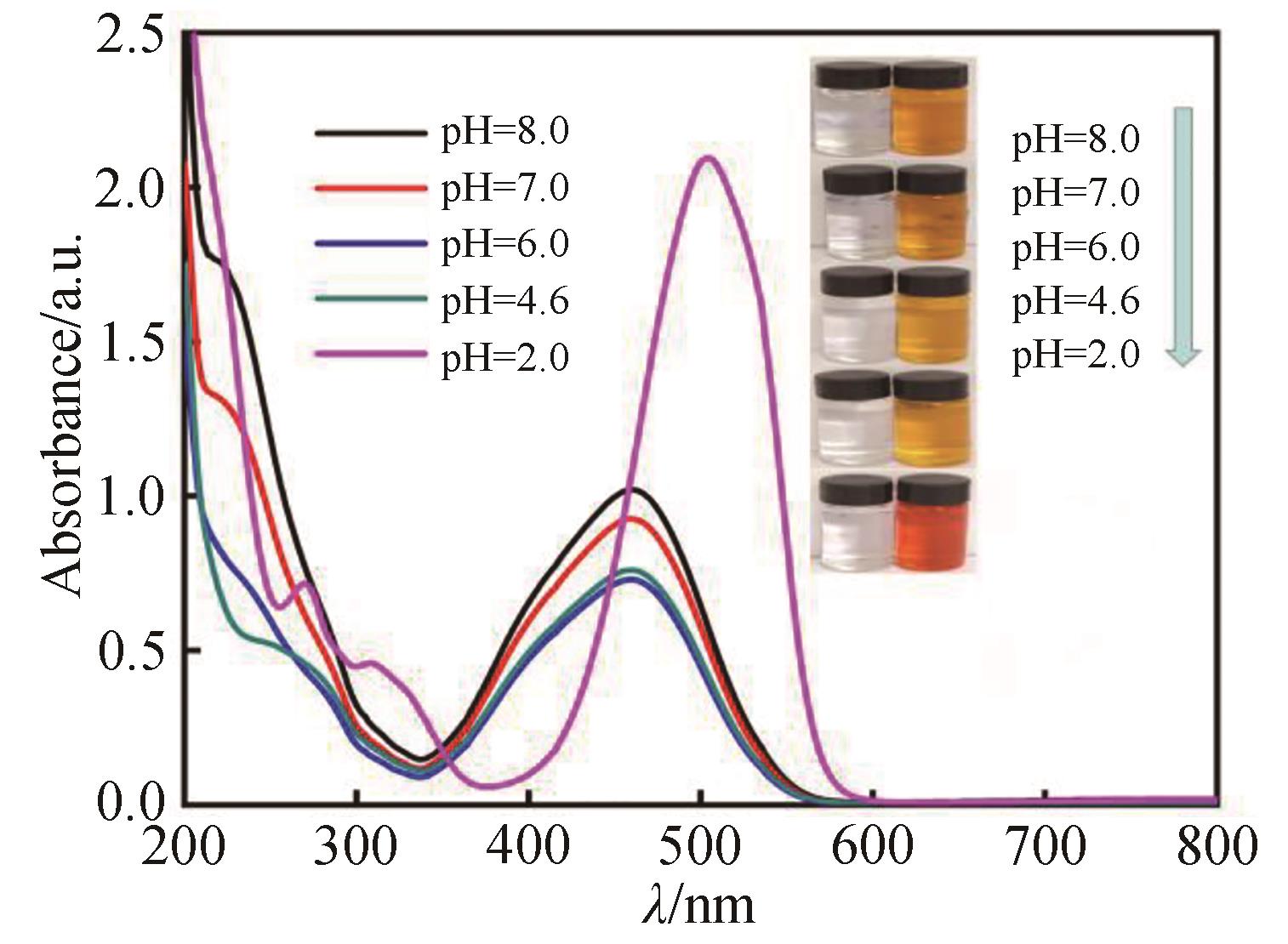
Fig.9 UV-Vis spectra and photos (inset) for MO desorption with different pH (20 mg adsorbent saturated with MO added into 100 mL PBS, magnetic stirring rate: 1400 r/min)
| 1 | GUSAIN R, KUMAR N, RAY S S, et al. Recent advances in carbon nanomaterial-based adsorbents for water purification[J]. Coord Chem Rev, 2020, 405: 213111. |
| 2 | SUN W H, LIU D F, ZHANG M H, et al. Application of electrode materials and catalysts in electrocatalytic treatment of dye wastewater[J]. Frontiers Chem Sci Eng, 2021, 15(6): 1427-1443. |
| 3 | FRAGA L E, FRANCO J H, ORLANDI M O, et al. Photoelectrocatalytic oxidation of hair dye basic red 51 at W/WO3/TiO2 bicomposite photoanode activated by ultraviolet and visible radiation[J]. J Environ Chem Eng, 2013, 1(3): 194-199. |
| 4 | AREMU O H, AKINTAYO C O, NAIDOO E B, et al. Synthesis and applications of nano-sized zinc oxide in wastewater treatment: a review[J]. Int J Environ Sci Technol, 2021, 18(10): 3237-3256. |
| 5 | HUSSAIN S, ANEGGI E, GOI D, et al. Catalytic activity of metals in heterogeneous Fenton-like oxidation of wastewater contaminants: a review[J]. Environ Chem Lett, 2021, 19(3): 2405-2424. |
| 6 | MENG W Q, WANG G H, ZHANG M, et al. Generation of acid-base by bipolar membrane electrodialysis process during desalination of pesticide containing wastewater[J]. Desalin Water Treat, 2021, 217: 91-100. |
| 7 | BERA S P, GODHANIYA M, KOTHARI C, et al. Emerging and advanced membrane technology for wastewater treatment: a review[J]. J Basic Microb, 2022, 62(3/4): 245-259. |
| 8 | BAGHERI H, HONARVAR B, ABBASI M, et al. Experimental evaluation of Farashband gas refinery wastewater treatment through activated carbon and natural zeolite adsorption process[J]. Desalin Water Treat, 2021, 225: 190-202. |
| 9 | JIANG S, XI J F, DAI H Q, et al. Multifunctional cellulose paper-based materials and their application in complex wastewater treatment[J]. Int J Biol Macromol, 2022, 207: 414-423. |
| 10 | FENG S, YANG W B, ZHANG L J, et al. Casein-hydroxyapatite composite microspheres for strontium-containing wastewater treatment[J]. ACS ES&T Water, 2021, 1(4): 900-909. |
| 11 | TANG Y, LI Y, ZHAN L, et al. Removal of emerging contaminants (bisphenol A and antibiotics) from kitchen wastewater by alkali-modified biochar[J]. Sci Total Environ, 2022, 805: 150158. |
| 12 | HAN D X, ZHAO H J, GAO L L, et al. Preparation of carboxymethyl chitosan/phytic acid composite hydrogels for rapid dye adsorption in wastewater treatment[J]. Colloids Surf A: Physicochem Eng Aspects, 2021, 628: 127355. |
| 13 | HUANG X S, YIN R, QIAN L, et al. Processing conditions dependent tunable negative permittivity in reduced graphene oxide-alumina nanocomposites[J]. Ceram Int, 2019, 45(14): 17784-17792. |
| 14 | CHEN Y, GAO X, LIU X W, et al. Water collection from air by ionic liquids for efficient visible-light-driven hydrogen evolution by metal-free conjugated polymer photocatalysts[J].Renew Ener, 2020, 147: 594-601. |
| 15 | TRUONG H B, BAE S, CHO J, et al. Advances in application of g-C3N4-based materials for treatment of polluted water and wastewater via activation of oxidants and photoelectrocatalysis: a comprehensive review[J]. Chemosphere, 2022, 286: 131737. |
| 16 | LU X L, XU K, CHEN P Z, et al. Facile one step method realizing scalable production of g-C3N4 nanosheets and study of their photocatalytic H2 evolution activity[J]. J Mater Chem A, 2014, 2(44): 18924-18928. |
| 17 | ZHANG S T, LI G Q, DUAN L Y, et al. g-C3N4 synthesized from NH4SCN in a H2 atmosphere as a high performance photocatalyst for blue light-driven degradation of rhodamine B[J]. Rsc Adv, 2020, 10(33): 19669-19685. |
| 18 | MA D N, LI X M, WANG X Q, et al. Preparation of g-C3N4 Nanosheets/CuO with enhanced catalytic activity on the thermal decomposition of ammonium perchlorate[J]. Eur J Inorg Chem, 2021, 2021(10): 982-988. |
| 19 | WU L F, ZHANG Z Z, YANG M M, et al. Facile synthesis of CuO/g-C3N4 hybrids for enhancing the wear resistance of polyimide composite[J]. Eur Polym J, 2019, 116: 463-470. |
| 20 | WANG Q Y, XU H L, HUANG W T, et al. Metal organic frameworks-assisted fabrication of CuO/Cu2O for enhanced selective catalytic reduction of NOx by NH3 at low temperatures[J]. J Hazard Mater, 2019, 364: 499-508. |
| 21 | ZAYED A M, WAHED M, MOHAMED E A, et al. Insights on the role of organic matters of some Egyptian clays in methyl orange adsorption: isotherm and kinetic studies[J]. Appl Clay Sci, 2018, 166: 49-60. |
| 22 | LIU Y, WU Y H, PANG H W, et al. Study on the removal of water pollutants by graphite phase carbon nitride materials[J]. Prog Chem, 2019, 31(6): 831-846. |
| 23 | WANG W, CAI K, WU X F, et al. A novel poly(m-phenylenediamine)/reduced graphene oxide/nickel ferritemagnetic adsorbent with excellent removal ability of dyes and Cr(VI)[J]. J Alloy Compd, 2017, 722: 532-543. |
| 24 | KANG D J, YU X L, GE M F, et al. Novel Al-doped carbon nanotubes with adsorption and coagulation promotion for organic pollutant removal[J]. J Environ Sci, 2017, 54: 1-12. |
| 25 | IMAN K, SHAHID M, KHAN M S, et al. Topology magnetism and dye adsorption properties of metal organic frameworks (MOFs) synthesized from bench chemicals[J]. CrystEngComm, 2019, 21(35): 5299-5309. |
| 26 | ZAYED A M, WAHED M, MOHAMED E A, et al. Insights on the role of organic matters of some Egyptian clays in methyl orange adsorption: isotherm and kinetic studies[J]. Appl Clay Sci, 2018, 166: 49-60. |
| 27 | GENG J Y, SI L L, GUO H T, et al. 3D nitrogen-doped graphene gels as robust and sustainable adsorbents for dyes[J]. New J Chem, 2017, 41(24): 15447-15457. |
| 28 | MINITHA C R, LALITHA M, JEYACHANDRAN Y L, et al. Adsorption behaviour of reduced graphene oxide towards cationic and anionic dyes: co-action of electrostatic and π-π interactions[J]. Mater Chem Phys, 2017, 194: 243-252. |
| [1] | LI Wenjin, YAO Weilong, XU Jiaxin, JIANG Qiying, DENG Hongquan. Preparation and Photocatalytic Properties of Ho3+ Doping BiFeO3 [J]. Chinese Journal of Applied Chemistry, 2019, 36(1): 91-96. |
| [2] | YU Zonglong, JIA Chunxiao, LU Xin, HAN Haigang, XIN Bingwei. Synthesis of Ionic Liquid@TiO2 Nanocomposites and Their Photocatalytic Performance [J]. Chinese Journal of Applied Chemistry, 2016, 33(4): 459-465. |
| [3] | XU Hui, TANG Jin, LI Chunlei, CHEN Yong. Polyaniline/attapulgite-supported Nanoscale Zero-valent Iron for Removing Methyl Orange in Aqueous Solution [J]. Chinese Journal of Applied Chemistry, 2016, 33(1): 92-97. |
| [4] | WANG Ping, GUO Xiangfeng*, JIA Lihua, MA Chunmiao, QIAN Lin. Interactions Between 1-Dodecyl-3-methylimidazolium Bromine and Methyl Orange [J]. Chinese Journal of Applied Chemistry, 2013, 30(07): 840-845. |
| [5] | ZHANG Feng, ZHANG Heng*, ZHU Wancheng, LI Yanxin, ZHAO Bin. Microwave Preparation of Fe-Pillared Bentonite and its Photocatalytic Activity for the Degradation of Methyl Orange [J]. Chinese Journal of Applied Chemistry, 2012, 29(06): 668-673. |
| [6] | MENG Dan, WANG Heyi*, LIU Xiuhua, DING Lanlan. Effects of Fe-Doping on the Structures and Photocatalytic Activities of TiO2 Thin Films [J]. Chinese Journal of Applied Chemistry, 2011, 28(12): 1379-1386. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||