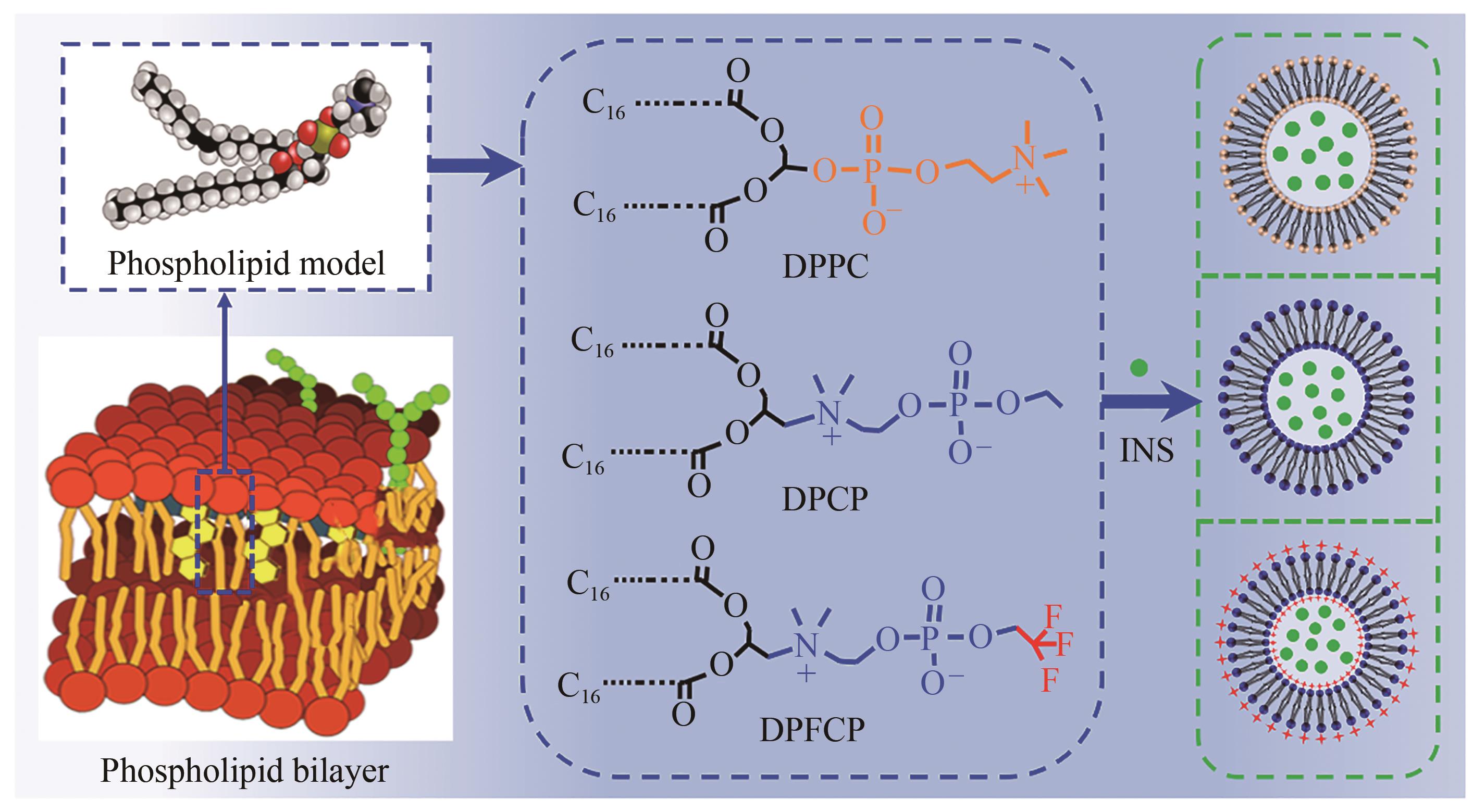
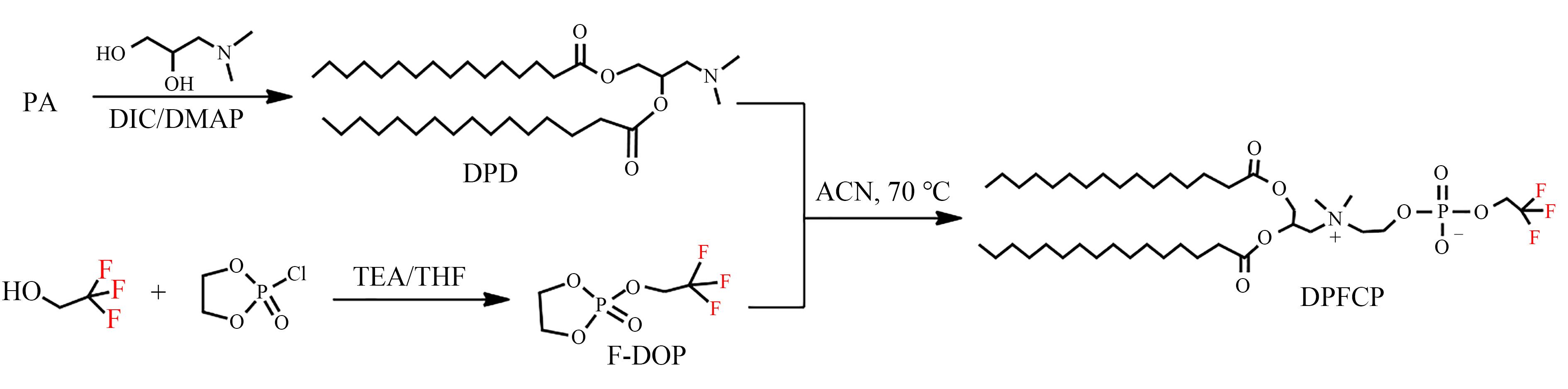
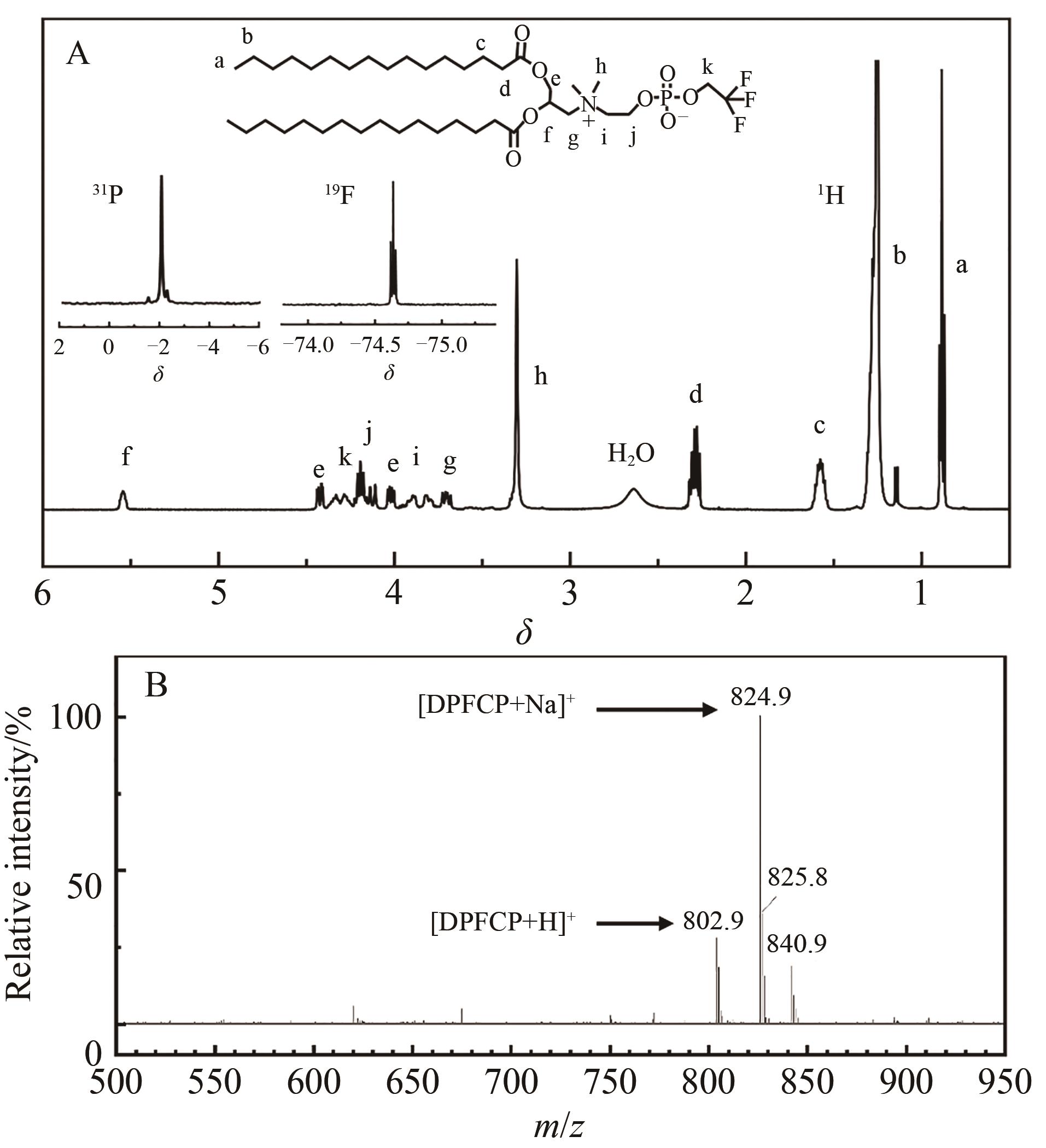
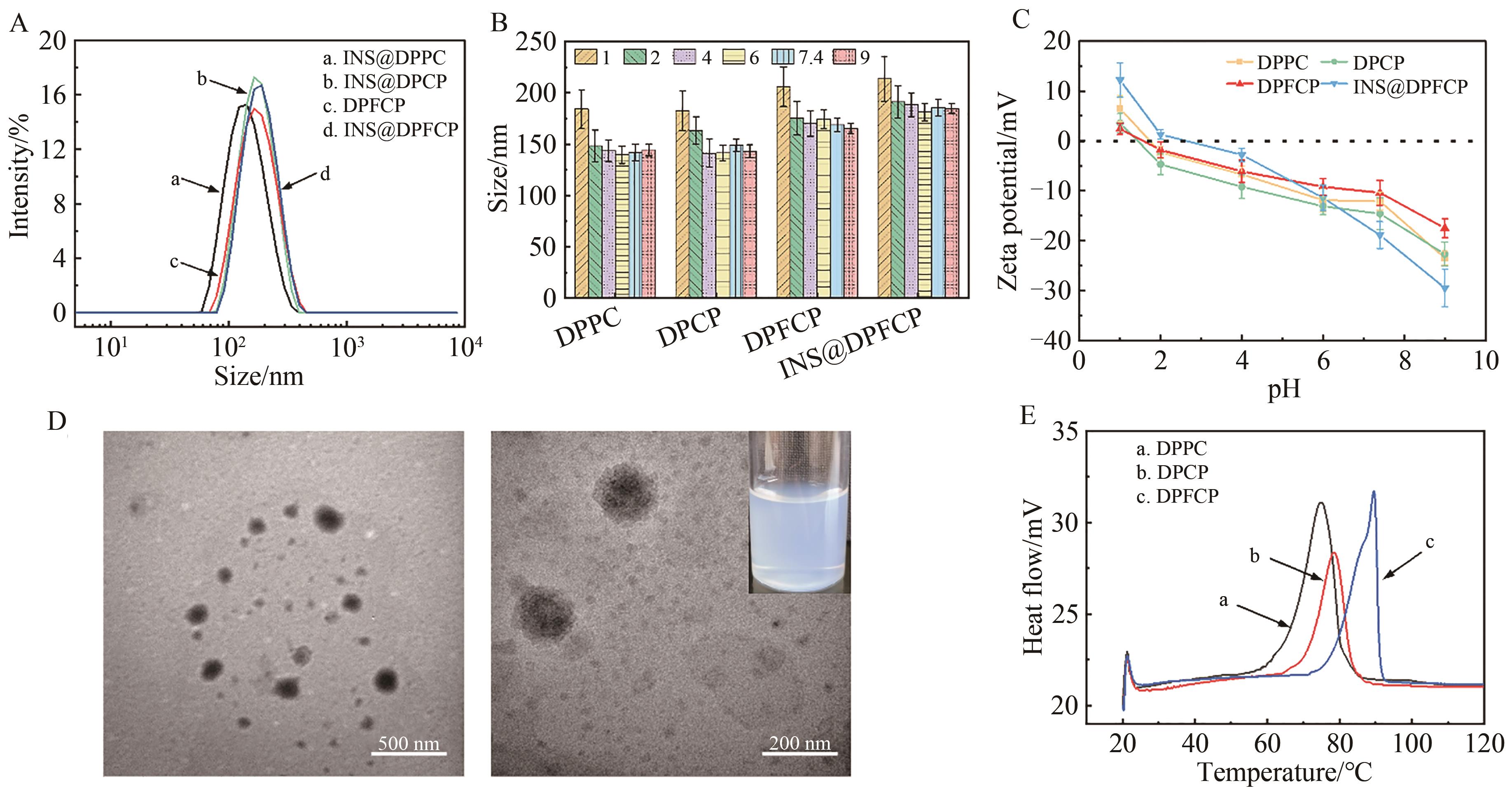
| 1 |
HEALD A H, STEDMAN M, DAVIES M, et al. Estimating life years lost to diabetes: outcomes from analysis of national diabetes audit and office of national statistics data[J]. Cardiovasc Endocr Me, 2020, 9(4): 183-185.
|
| 2 |
SUN H, SAEEDI P, KARURANGA S, et al. IDF diabetes atlas: global, regional and country-level diabetes prevalence estimates for 2021 and projections for 2045[J]. Diabetes Res Clin Pr, 2022, 183: 109119.
|
| 3 |
XIAO Y F, WANG J Q, LIU C, et al. Oral insulin delivery platforms: strategies to address the biological barriers[J]. Angew Chem Int Ed, 2020, 59(45): 19787-19795.
|
| 4 |
AMRITA B, KELLY L, TYLER B, et al. Ionic liquids for oral insulin delivery[J]. PNAS, 2018, 115(28): 7296-7301.
|
| 5 |
Al-REMAWI M, ELSAYED A, MAGHRABI I, et al. Chitosan/lecithin liposomal nanovesicles as an oral insulin delivery system[J]. Pharm Dev Technol, 2016, 22(3): 390-398.
|
| 6 |
MOROZ E, MATOORI S, LEROUX J C, et al. Oral delivery of macromolecular drugs: where we are after almost 100 years of attempts[J]. Adv Drug Delivery Rev, 2016, 101: 108-121.
|
| 7 |
ZHU Q G, CHEN Z J, PAUL P K, et al. Oral delivery of proteins and peptides: challenges, status quo and future perspectives[J]. Acta Pharm Sin B, 2021, 11(8): 2416-2448.
|
| 8 |
黄琼卫, 赵丽, 宋之明, 等. 葡萄糖敏感苯硼酸基高分子纳米药物传输体系[J]. 应用化学, 2017, 34(7): 733-743.
|
|
HUANG Q W, ZHAO L, SONG Z M, et al. Phenylboronic acid-based glucose-sensitive polymer nanocarriers used as drug delivery systems[J]. Chin J Appl Chem, 2017, 34(7): 733-743.
|
| 9 |
YUN Y, CHO Y W, PARK K. Nanoparticles for oral delivery: targeted nanoparticles with peptidic ligands for oral protein delivery[J]. Adv Drug Delivery Rev, 2013, 65(6): 822-832.
|
| 10 |
ZUBEN E S V, ELOY J O, ARAUJO V H S, et al. Insulin-loaded liposomes functionalized with cell-penetrating peptides: influence on drug release and permeation through porcine nasal mucosa[J]. Colloids Surf A, 2021, 622: 126624.
|
| 11 |
JASH A, UBEYITOGULLARI A, RIZVI S S H. Liposomes for oral delivery of protein and peptide-based therapeutics: challenges, formulation strategies, and advances[J]. J Mater Chem B, 2021, 9(24): 4773-4792.
|
| 12 |
韩旭, 丁冠宇, 董青, 等. 基于脂质体的纳米基因载体的研究进展[J]. 应用化学, 2018, 35(7): 735-744.
|
|
HAN X, DING G Y, DONG Q, et al. Research progress of nano-gene carriers based on liposomes[J]. Chin J Appl Chem, 2018, 35(7): 735-744.
|
| 13 |
HAN X, LU Y, XIE J, et al. Zwitterionic micelles efficiently deliver oral insulin without opening tight junctions[J]. Nat Nanotechnol, 2020, 15(7): 605-614.
|
| 14 |
HU W Y, WU Z M, YANG Q Q, et al. Smart pH-responsive polymeric micelles for programmed oral delivery of insulin[J]. Colloids Surf B, 2019, 183: 110443.
|
| 15 |
ZENG Z Y, QI D M, YANG L, et al. Stimuli-responsive self-assembled dendrimers for oral protein delivery[J]. J Controlled Release, 2019, 315: 206-213.
|
| 16 |
WEN N, LÜ S Y, GAO C M, et al. Glucose-responsive zwitterionic dialdehyde starch-based micelles with potential anti-phagocytic behavior for insulin delivery[J]. Chem Eng J, 2018, 335: 52-62.
|
| 17 |
RAO R, LIU X H, LI Y H, et al. Bioinspired zwitterionic polyphosphoester modified porous silicon nanoparticles for efficient oral Insulin delivery[J]. Biomater Sci, 2021, 9(3): 685-699.
|
| 18 |
LI Y, JI W H, PENG H, et al. Charge-switchable zwitterionic polycarboxybetaine particle as an intestinal permeation enhancer for efficient oral insulin delivery[J]. Theranostics, 2021, 11(9): 4452-4466.
|
| 19 |
ZHOU Y H, LIU L, CAO Y, et al. A nanocomposite vehicle based on metal-organic framework nanoparticle incorporated biodegradable microspheres for enhanced oral insulin delivery[J]. ACS Appl Mater Interfaces, 2020, 12(20): 22581-22592.
|
| 20 |
HE M Y, PEI Y, HU Y L, et al. Erythrocyte-membrane-enveloped biomineralized metal-organic framework nanoparticles enable intravenous glucose-responsive insulin delivery[J]. ACS Appl Mater Interfaces, 2021, 13(17): 19648-19659.
|
| 21 |
LIU L, ZHANG Y, YU S J, et al. pH-and amylase-responsive carboxymethyl starch/poly(2-isobutyl-acrylic acid) hybrid microgels as effective enteric carriers for oral insulin delivery[J]. Biomacromolecules, 2018, 19(6): 2123-2136.
|
| 22 |
HU Y W, GAO S J, LU H F, et al. Acid-resistant and physiological pH-responsive DNA hydrogel composed of a-motif and i-motif toward oral insulin delivery[J]. J Am Chem Soc, 2022, 144(12): 5461-5470.
|
| 23 |
HE H S, LU Y, QI J P, et al. Adapting liposomes for oral drug delivery[J]. Acta Pharm Sin B, 2019, 9(1): 36-48.
|
| 24 |
VITALE A, BONGIOVANNI R, AMEDURI B. Fluorinated oigomers and polymers in photopolymerization[J]. Chem Rev, 2015, 115(16): 8835-8866.
|
| 25 |
CAO Y, WU H P, ALLEC S I, et al. A highly stretchy, transparent elastomer with the capability to automatically self-heal underwater[J]. Adv Mater, 2018, 30(49): 1804602.
|
| 26 |
侯成敏, 李娜, 董海涛, 等. 含氟环氧树脂杂化纳米二氧化硅超疏水材料的制备与性能[J]. 应用化学, 2019, 36(7): 798-806.
|
|
HOU C M, LI N, DONG H T, et al. Preparation and performance of hybrid superhydrophobic materials from fluorinated epoxy resin and silica nanoparticles[J]. Chin J Appl Chem, 2019, 36(7): 798-806.
|
| 27 |
WU P K, ZHANG H T, SUN M L, et al. Manipulating offense and defense signaling to fight cold tumors with carrier-free nanoassembly of fluorinated prodrug and siRNA[J]. Adv Mater, 2022, 34(38): 2203019.
|
| 28 |
LI G Z, WANG S P, DENG D S, et al. Fluorinated chitosan to enhance transmucosal delivery of sonosensitizer-conjugated catalase for sonodynamic bladder cancer treatment post-intravesical instillation[J]. ACS Nano, 2020, 14(2): 1586-1599.
|
| 29 |
LI G Z, LEI Q F, WANG F, et al. Fluorinated polymer mediated transmucosal peptide delivery for intravesical instillation therapy of bladder cancer[J]. Small, 2019, 15(25): 1900936.
|
| 30 |
LI S R, MEI W K, WANG X Z, et al. Choline phosphate lipid insertion and rigidification of cell membranes for targeted cancer chemo-immunotherapy[J]. Chem Commun, 2021, 57: 1372-1375.
|
| 31 |
WANG W L, JIANG S N, LI S R, et al. Functional choline phosphate lipids for enhanced drug delivery in cancer therapy[J]. Chem Mater, 2021, 33(2): 774-781.
|
| 32 |
ZHANG X W, QI J P, LU Y, et al. Biotinylated liposomes as potential carriers for the oral delivery of insulin[J]. Nanomed: Nanotechnol Biol Med, 2014, 10(1): 167-176.
|
| 33 |
NIU M M, LU Y, HOVGAARD L, et al. Hypoglycemic activity and oral bioavailability of insulin-loaded liposomes containing bile salts in rats: the effect of cholate type, particle size and administered dose[J]. Eur J Pharm Biopharm, 2012, 81(2): 265-272.
|
| 34 |
ZHOU Y H, CHEN Z X, CHEN X S, et al. A pH-triggered self-unpacking capsule containing zwitterionic hydrogel-coated MOF nanoparticles for efficient oral exendin-4 delivery[J]. Adv Mater, 2021, 33(32): 2102044.
|
| 35 |
WONG C Y, AL-SALAMI H, DASS C R, et al. Recent advancements in oral administration of insulin-loaded liposomal drug delivery systems for diabetes mellitus[J]. Int J Pharmaceut, 2018, 549: 201-217.
|
| 36 |
ZHU X, WU J, SHAN W, et al. Sub-50 nm nanoparticles with biomimetic surfaces to sequentially overcome the mucosal diffusion barrier and the epithelial absorption barrier[J]. Adv Funct Mater, 2016, 26(16): 2728-2738.
|
| 37 |
PRESAS E, MCCARTNEY F, SULTAN E, et al. Physicochemical, pharmacokinetic and pharmacodynamic analyses of amphiphilic cyclodextrin-based nanoparticles designed to enhance intestinal delivery of insulin[J]. J Controlled Release, 2018, 286: 402-414.
|
| 38 |
MAGARKAR A, RÓG T, BUNKER A. Molecular dynamics simulation of inverse-phosphocholine lipids[J]. J Phys Chem C, 2014, 118(33): 19444-19449.
|
| 39 |
ZHANG C, LIU T Q, WANG W Q, et al. Tuning of the aggregation behavior of fluorinated polymeric nanoparticles for improved therapeutic efficacy[J]. ACS Nano, 2020, 14(6): 7425-7434.
|
| 40 |
CHEN X Y, CHEN T C, LIN Z F, et al. Choline phosphate functionalized surface: protein-resistant but cell-adhesive zwitterionic surface potential for tissue engineering[J]. Chem Commun, 2015, 51(3): 487-490.
|

 )
)