Chinese Journal of Applied Chemistry ›› 2024, Vol. 41 ›› Issue (3): 437-444.DOI: 10.19894/j.issn.1000-0518.230355
• Full Papers • Previous Articles Next Articles
Fingerprint Construction and Identification of Common Peaks for Tenghuang Jiangu Wan Based on UPLC-Q TOF-MSE
Dong XIE1, Jian-Nan HU1, Nian LI1, Ju YANG2, Qing HUANG2, Zi-Feng PI1( ), Fei ZHENG1, Yu-Lin DAI1, Hao YUE1(
), Fei ZHENG1, Yu-Lin DAI1, Hao YUE1( )
)
- 1.Jilin Ginseng Acadmg,Changchun University of Chinese Medicine,Changchun 130117,China
2.Changchun People Pharmaceutical Group Co. ,Ltd. ,Changchun 130033,China
-
Received:2023-11-10Accepted:2024-03-01Published:2024-03-01Online:2024-04-09 -
Contact:Zi-Feng PI,Hao YUE -
Supported by:Science and Technology Development Project of Jilin Province(20210401064YY)
CLC Number:
Cite this article
Dong XIE, Jian-Nan HU, Nian LI, Ju YANG, Qing HUANG, Zi-Feng PI, Fei ZHENG, Yu-Lin DAI, Hao YUE. Fingerprint Construction and Identification of Common Peaks for Tenghuang Jiangu Wan Based on UPLC-Q TOF-MSE[J]. Chinese Journal of Applied Chemistry, 2024, 41(3): 437-444.
share this article
Add to citation manager EndNote|Ris|BibTeX
URL: http://yyhx.ciac.jl.cn/EN/10.19894/j.issn.1000-0518.230355
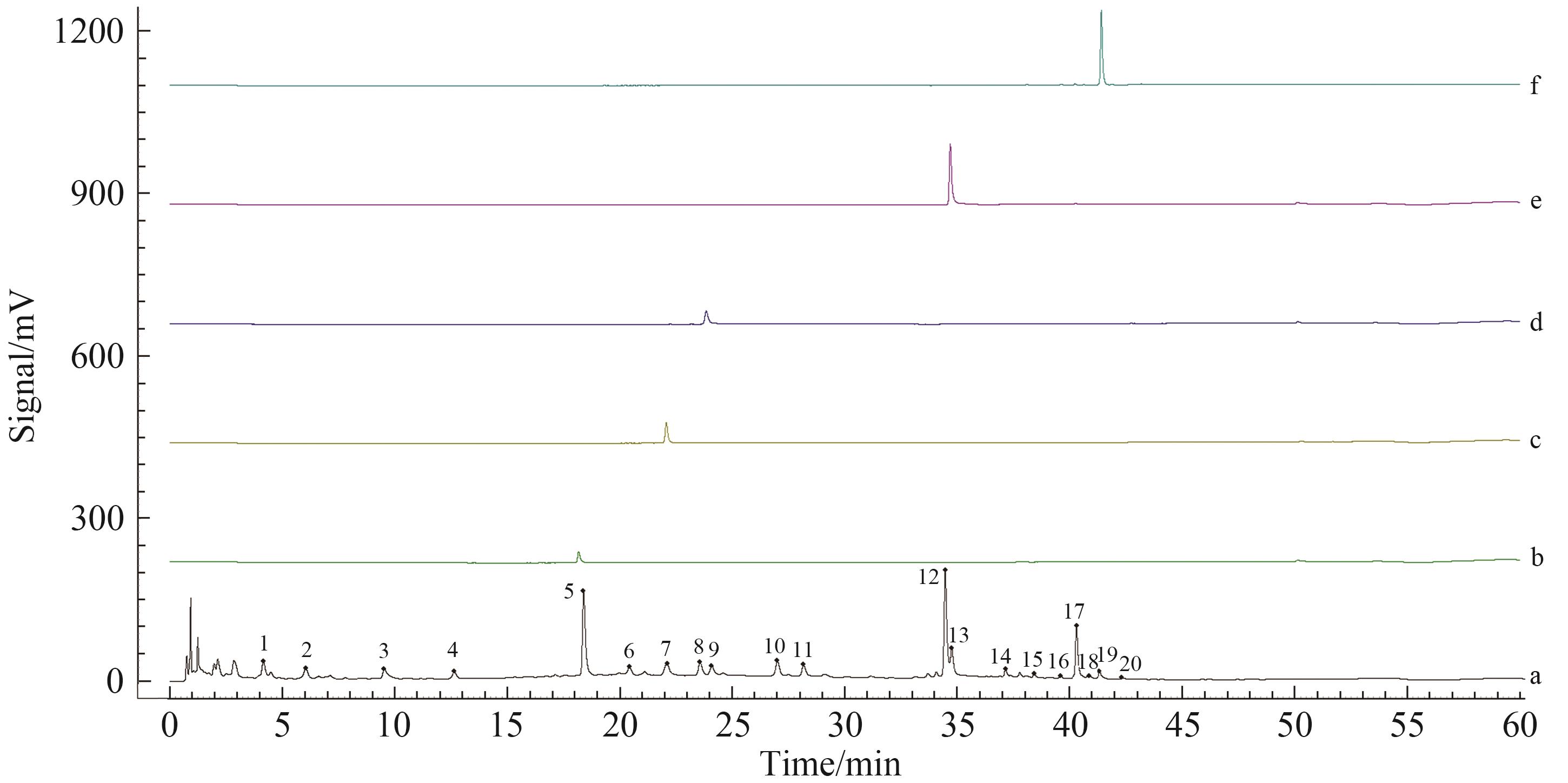
Fig. 2 Reference fingerprints (a) and HPLC chromatograms of reference standards (b. Echinacoside; c. Acteoside; d. Naringin; e. Icariin; f. Icariside Ⅱ)
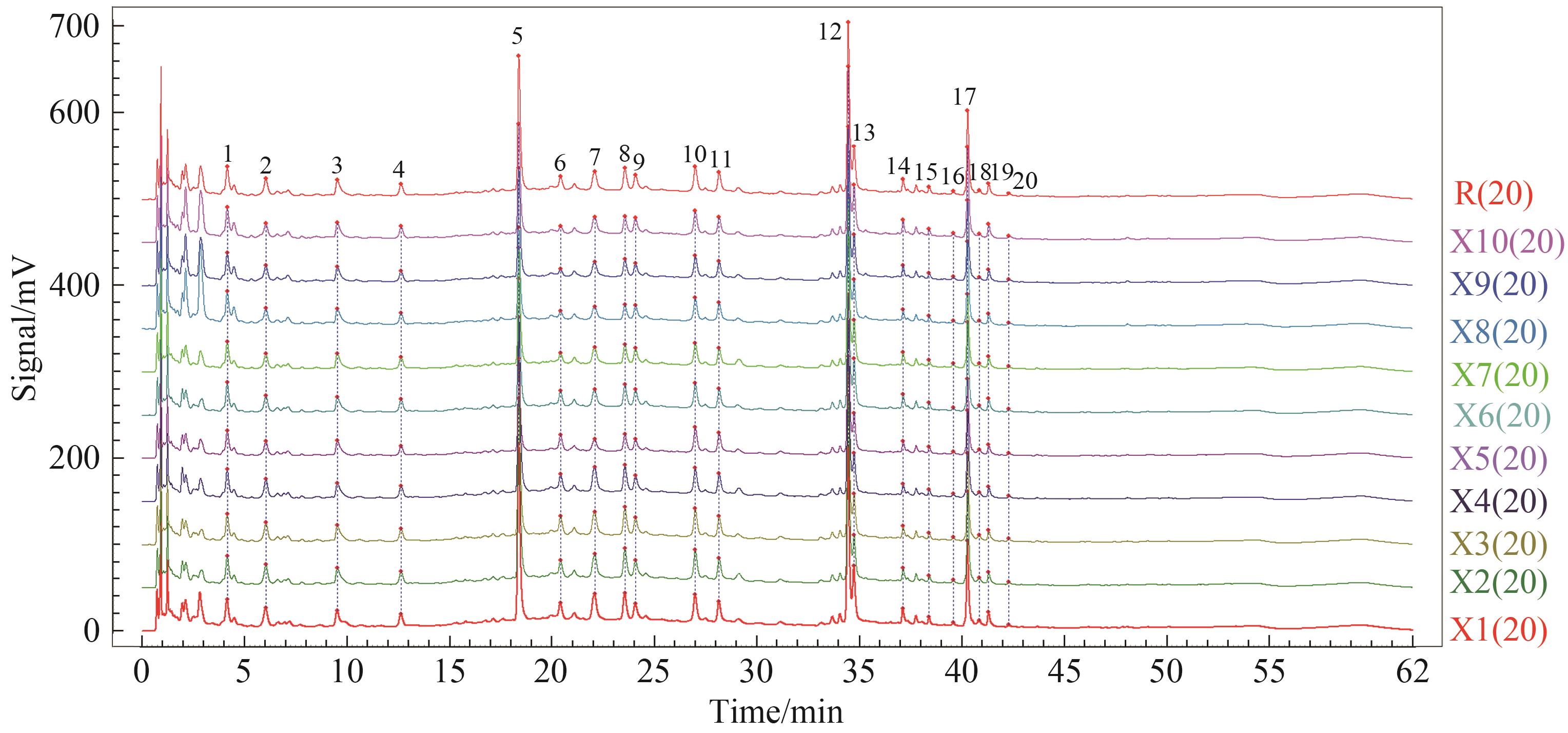
Peaks (No.) | Retention time/min | Selected ion | Observed(m/z) | Mass error/(mg·L-1) | Major product ions (m/z) | Compound | Attribution |
|---|---|---|---|---|---|---|---|
| 1 | 4.13 | [M-H]- | 331.103 8 | 1.0 | 179.056 0 | Koaburaside | a |
| 2 | 5.95 | [M-H]- | 595.166 5 | -0.7 | 459.113 7, 235.024 5 | Neoeriocitrin | b |
| 3 | 9.12 | [M+H]+ | 825.278 9 | -2.8 | 663.229 9, 517.170 4, 355.118 0 | Diphylloside A | c |
| 5 | 18.37 | [M-H]- | 785.250 6 | -0.4 | 623.218 8, 461.166 0, 179.034 7 | Echinacoside | d |
| 7 | 22.14 | [M-H]- | 623.197 6 | -0.9 | 461.164 9, 315.104 8, 179.035 5 | Acteoside | d,e |
| 8 | 23.61 | [M-H]- | 579.171 2 | -1.2 | 271.060 7, 151.003 3 | Naringin | b |
| 9 | 24.14 | [M+H]+ | 273.075 9 | 0.6 | 153.018 1, 119.049 2 | Naringenin | b |
| 10 | 27.06 | [M-H]- | 753.224 8 | 0.2 | 223.060 4, 205.050 3, 190.026 6 | 3,6-Disinapoyl sucrose | f |
| 11 | 28.23 | [M+HCOO]- | 853.276 7 | -0.6 | 645.218 2, 366.110 4, 351.087 0 | Epimedin B | c |
| 12 | 34.51 | [M+HCOO]- | 721.234 3 | -0.8 | 513.175 9, 367.117 5, 351.086 6 | Icariin | c |
| 13 | 34.81 | [M-H]- | 659.233 5 | -1.6 | 367.116 5, 351.086 9, 323.092 0 | 2-O-Rhamnosylicariside Ⅱ | c |
| 17 | 40.36 | [M-H]- | 529.170 6 | -1.8 | 367.117 7, 352.092 9 | Icariside Ⅰ | c |
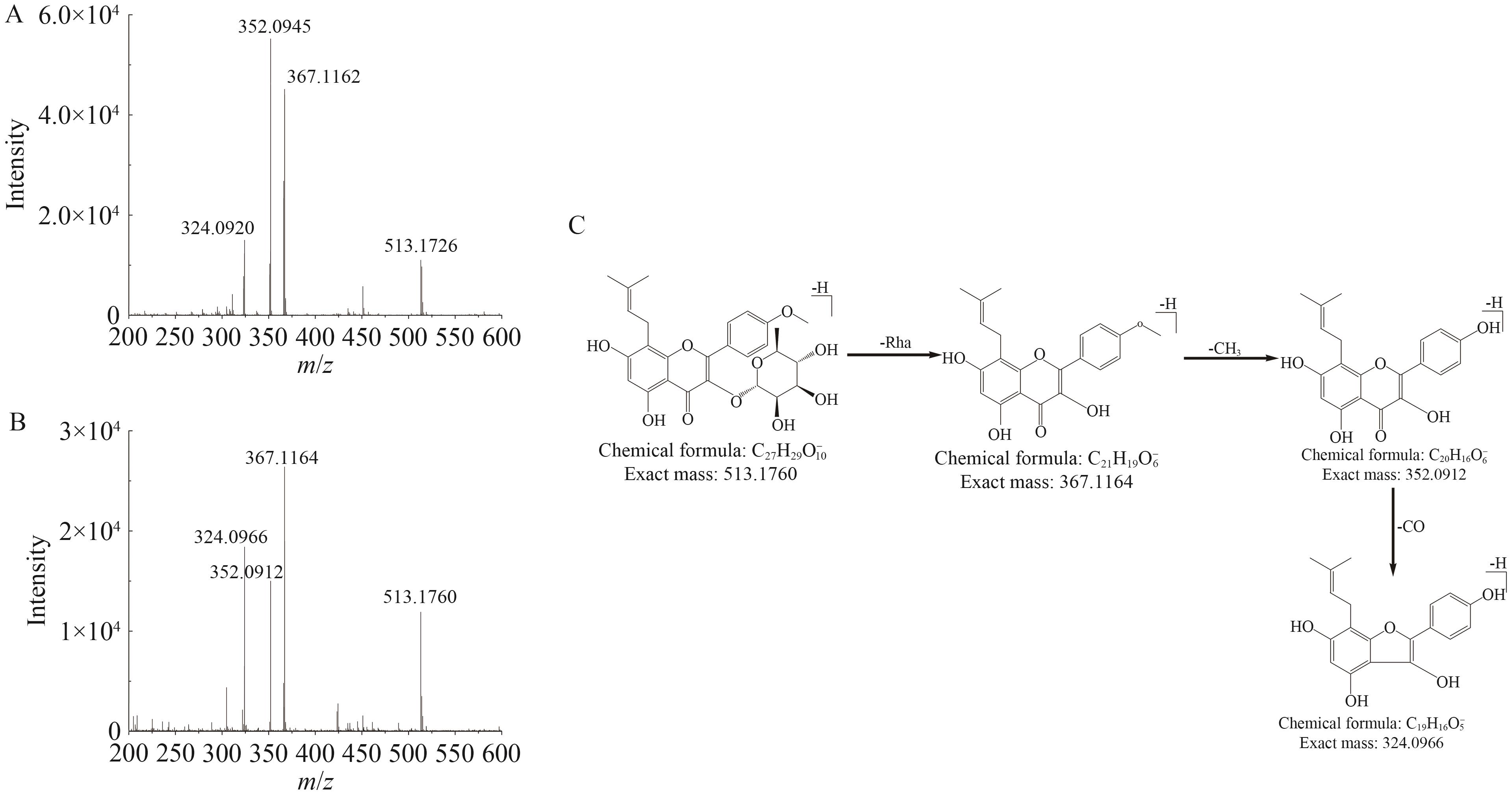
| 19 | 41.39 | [M-H]- | 513.176 0 | -1.2 | 367.116 4, 352.091 2, 324.096 6 | Icariside Ⅱ | c |
Table 1 Identification of 13 common peaks by mass spectrometry
Peaks (No.) | Retention time/min | Selected ion | Observed(m/z) | Mass error/(mg·L-1) | Major product ions (m/z) | Compound | Attribution |
|---|---|---|---|---|---|---|---|
| 1 | 4.13 | [M-H]- | 331.103 8 | 1.0 | 179.056 0 | Koaburaside | a |
| 2 | 5.95 | [M-H]- | 595.166 5 | -0.7 | 459.113 7, 235.024 5 | Neoeriocitrin | b |
| 3 | 9.12 | [M+H]+ | 825.278 9 | -2.8 | 663.229 9, 517.170 4, 355.118 0 | Diphylloside A | c |
| 5 | 18.37 | [M-H]- | 785.250 6 | -0.4 | 623.218 8, 461.166 0, 179.034 7 | Echinacoside | d |
| 7 | 22.14 | [M-H]- | 623.197 6 | -0.9 | 461.164 9, 315.104 8, 179.035 5 | Acteoside | d,e |
| 8 | 23.61 | [M-H]- | 579.171 2 | -1.2 | 271.060 7, 151.003 3 | Naringin | b |
| 9 | 24.14 | [M+H]+ | 273.075 9 | 0.6 | 153.018 1, 119.049 2 | Naringenin | b |
| 10 | 27.06 | [M-H]- | 753.224 8 | 0.2 | 223.060 4, 205.050 3, 190.026 6 | 3,6-Disinapoyl sucrose | f |
| 11 | 28.23 | [M+HCOO]- | 853.276 7 | -0.6 | 645.218 2, 366.110 4, 351.087 0 | Epimedin B | c |
| 12 | 34.51 | [M+HCOO]- | 721.234 3 | -0.8 | 513.175 9, 367.117 5, 351.086 6 | Icariin | c |
| 13 | 34.81 | [M-H]- | 659.233 5 | -1.6 | 367.116 5, 351.086 9, 323.092 0 | 2-O-Rhamnosylicariside Ⅱ | c |
| 17 | 40.36 | [M-H]- | 529.170 6 | -1.8 | 367.117 7, 352.092 9 | Icariside Ⅰ | c |
| 19 | 41.39 | [M-H]- | 513.176 0 | -1.2 | 367.116 4, 352.091 2, 324.096 6 | Icariside Ⅱ | c |
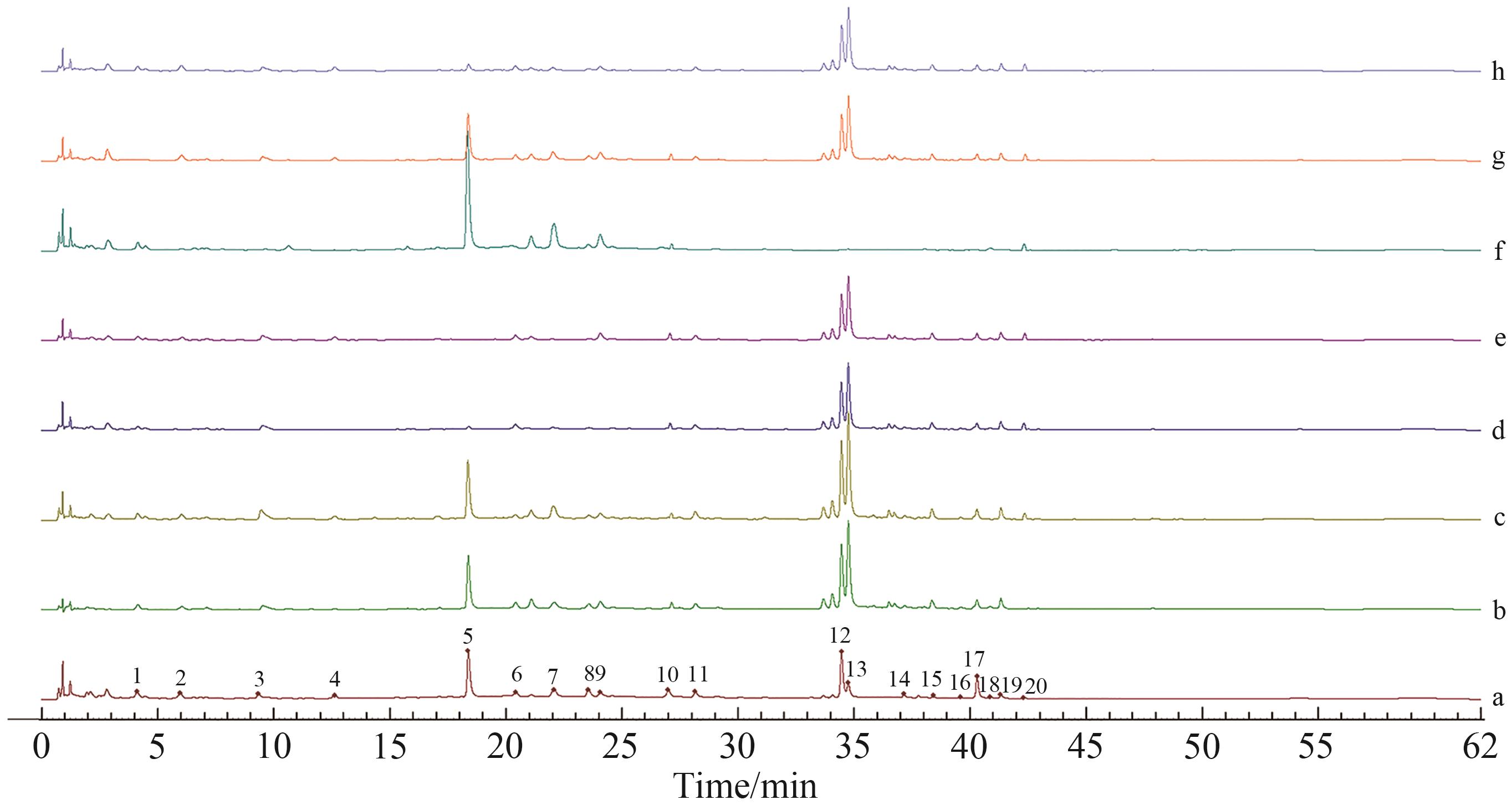
Fig.4 Negative controls of fingerprintsNote: a. The test solution; b. Negative sample solution of Radix Rehmanniae Praeparata; c. Negative sample solution of Pyrola; d. Negative sample solution of Rhizoma Drynariae; e. Negative sample solution of Desertliving Cistanche; f. Negative sample solution of Epimedium brevicornum; g. Negative sample solution of Spatholobus; h. Negative sample solution of Radish Seed
| Batch No. | X1 | X2 | X3 | X4 | X5 | X6 | X7 | X8 | X9 | X10 | R |
|---|---|---|---|---|---|---|---|---|---|---|---|
| X1 | 1.000 | 0.998 | 0.997 | 0.998 | 0.959 | 0.982 | 0.980 | 0.959 | 0.979 | 0.967 | 0.991 |
| X2 | 0.998 | 1.000 | 1.000 | 0.999 | 0.968 | 0.987 | 0.986 | 0.966 | 0.984 | 0.973 | 0.995 |
| X3 | 0.997 | 1.000 | 1.000 | 0.999 | 0.970 | 0.987 | 0.987 | 0.967 | 0.984 | 0.973 | 0.995 |
| X4 | 0.998 | 0.999 | 0.999 | 1.000 | 0.964 | 0.984 | 0.983 | 0.962 | 0.980 | 0.969 | 0.992 |
| X5 | 0.959 | 0.968 | 0.970 | 0.964 | 1.000 | 0.994 | 0.994 | 0.996 | 0.993 | 0.994 | 0.988 |
| X6 | 0.982 | 0.987 | 0.987 | 0.984 | 0.994 | 1.000 | 0.999 | 0.992 | 0.998 | 0.995 | 0.998 |
| X7 | 0.980 | 0.986 | 0.987 | 0.983 | 0.994 | 0.999 | 1.000 | 0.992 | 0.997 | 0.995 | 0.997 |
| X8 | 0.959 | 0.966 | 0.967 | 0.962 | 0.996 | 0.992 | 0.992 | 1.000 | 0.996 | 0.998 | 0.987 |
| X9 | 0.979 | 0.984 | 0.984 | 0.980 | 0.993 | 0.998 | 0.997 | 0.996 | 1.000 | 0.998 | 0.997 |
| X10 | 0.967 | 0.973 | 0.973 | 0.969 | 0.994 | 0.995 | 0.995 | 0.998 | 0.998 | 1.000 | 0.991 |
| R | 0.991 | 0.995 | 0.995 | 0.992 | 0.988 | 0.998 | 0.997 | 0.987 | 0.997 | 0.991 | 1.000 |
Table 2 Fingerprint similarity calculation results of 10 batches finished products
| Batch No. | X1 | X2 | X3 | X4 | X5 | X6 | X7 | X8 | X9 | X10 | R |
|---|---|---|---|---|---|---|---|---|---|---|---|
| X1 | 1.000 | 0.998 | 0.997 | 0.998 | 0.959 | 0.982 | 0.980 | 0.959 | 0.979 | 0.967 | 0.991 |
| X2 | 0.998 | 1.000 | 1.000 | 0.999 | 0.968 | 0.987 | 0.986 | 0.966 | 0.984 | 0.973 | 0.995 |
| X3 | 0.997 | 1.000 | 1.000 | 0.999 | 0.970 | 0.987 | 0.987 | 0.967 | 0.984 | 0.973 | 0.995 |
| X4 | 0.998 | 0.999 | 0.999 | 1.000 | 0.964 | 0.984 | 0.983 | 0.962 | 0.980 | 0.969 | 0.992 |
| X5 | 0.959 | 0.968 | 0.970 | 0.964 | 1.000 | 0.994 | 0.994 | 0.996 | 0.993 | 0.994 | 0.988 |
| X6 | 0.982 | 0.987 | 0.987 | 0.984 | 0.994 | 1.000 | 0.999 | 0.992 | 0.998 | 0.995 | 0.998 |
| X7 | 0.980 | 0.986 | 0.987 | 0.983 | 0.994 | 0.999 | 1.000 | 0.992 | 0.997 | 0.995 | 0.997 |
| X8 | 0.959 | 0.966 | 0.967 | 0.962 | 0.996 | 0.992 | 0.992 | 1.000 | 0.996 | 0.998 | 0.987 |
| X9 | 0.979 | 0.984 | 0.984 | 0.980 | 0.993 | 0.998 | 0.997 | 0.996 | 1.000 | 0.998 | 0.997 |
| X10 | 0.967 | 0.973 | 0.973 | 0.969 | 0.994 | 0.995 | 0.995 | 0.998 | 0.998 | 1.000 | 0.991 |
| R | 0.991 | 0.995 | 0.995 | 0.992 | 0.988 | 0.998 | 0.997 | 0.987 | 0.997 | 0.991 | 1.000 |
| Batch No. | R | Z1 | Z2 | Z3 | Z4 | Z5 | Z6 | Z7 | Z8 | Z9 | Z10 |
|---|---|---|---|---|---|---|---|---|---|---|---|
| R | 1.000 | 0.975 | 0.981 | 0.985 | 0.981 | 0.995 | 0.998 | 0.998 | 0.995 | 0.998 | 0.997 |
| Z1 | 0.975 | 1.000 | 0.995 | 0.997 | 0.997 | 0.956 | 0.981 | 0.974 | 0.954 | 0.980 | 0.968 |
| Z2 | 0.981 | 0.995 | 1.000 | 0.999 | 0.999 | 0.967 | 0.988 | 0.983 | 0.961 | 0.984 | 0.971 |
| Z3 | 0.985 | 0.997 | 0.999 | 1.000 | 0.999 | 0.972 | 0.990 | 0.986 | 0.967 | 0.987 | 0.976 |
| Z4 | 0.981 | 0.997 | 0.999 | 0.999 | 1.000 | 0.965 | 0.987 | 0.981 | 0.961 | 0.984 | 0.972 |
| Z5 | 0.995 | 0.956 | 0.967 | 0.972 | 0.965 | 1.000 | 0.993 | 0.994 | 0.995 | 0.990 | 0.991 |
| Z6 | 0.998 | 0.981 | 0.988 | 0.990 | 0.987 | 0.993 | 1.000 | 0.997 | 0.991 | 0.997 | 0.993 |
| Z7 | 0.998 | 0.974 | 0.983 | 0.986 | 0.981 | 0.994 | 0.997 | 1.000 | 0.991 | 0.995 | 0.993 |
| Z8 | 0.995 | 0.954 | 0.961 | 0.967 | 0.961 | 0.995 | 0.991 | 0.991 | 1.000 | 0.994 | 0.997 |
| Z9 | 0.998 | 0.980 | 0.984 | 0.987 | 0.984 | 0.990 | 0.997 | 0.995 | 0.994 | 1.000 | 0.998 |
| Z10 | 0.997 | 0.968 | 0.971 | 0.976 | 0.972 | 0.991 | 0.993 | 0.993 | 0.997 | 0.998 | 1.000 |
Table 3 Calculated similarity results of intermediate-control fingerprint
| Batch No. | R | Z1 | Z2 | Z3 | Z4 | Z5 | Z6 | Z7 | Z8 | Z9 | Z10 |
|---|---|---|---|---|---|---|---|---|---|---|---|
| R | 1.000 | 0.975 | 0.981 | 0.985 | 0.981 | 0.995 | 0.998 | 0.998 | 0.995 | 0.998 | 0.997 |
| Z1 | 0.975 | 1.000 | 0.995 | 0.997 | 0.997 | 0.956 | 0.981 | 0.974 | 0.954 | 0.980 | 0.968 |
| Z2 | 0.981 | 0.995 | 1.000 | 0.999 | 0.999 | 0.967 | 0.988 | 0.983 | 0.961 | 0.984 | 0.971 |
| Z3 | 0.985 | 0.997 | 0.999 | 1.000 | 0.999 | 0.972 | 0.990 | 0.986 | 0.967 | 0.987 | 0.976 |
| Z4 | 0.981 | 0.997 | 0.999 | 0.999 | 1.000 | 0.965 | 0.987 | 0.981 | 0.961 | 0.984 | 0.972 |
| Z5 | 0.995 | 0.956 | 0.967 | 0.972 | 0.965 | 1.000 | 0.993 | 0.994 | 0.995 | 0.990 | 0.991 |
| Z6 | 0.998 | 0.981 | 0.988 | 0.990 | 0.987 | 0.993 | 1.000 | 0.997 | 0.991 | 0.997 | 0.993 |
| Z7 | 0.998 | 0.974 | 0.983 | 0.986 | 0.981 | 0.994 | 0.997 | 1.000 | 0.991 | 0.995 | 0.993 |
| Z8 | 0.995 | 0.954 | 0.961 | 0.967 | 0.961 | 0.995 | 0.991 | 0.991 | 1.000 | 0.994 | 0.997 |
| Z9 | 0.998 | 0.980 | 0.984 | 0.987 | 0.984 | 0.990 | 0.997 | 0.995 | 0.994 | 1.000 | 0.998 |
| Z10 | 0.997 | 0.968 | 0.971 | 0.976 | 0.972 | 0.991 | 0.993 | 0.993 | 0.997 | 0.998 | 1.000 |
| 1 | 张文静, 王雨辰, 段瑶, 等. 藤黄健骨丸调控BMP-2/Smad信号通路治疗绝经后骨质疏松症[J]. 吉林中医药, 2023, 43(12): 1459-1464. |
| ZHANG W J, WANG Y C, DUAN Y, et al. Effffect of Tenghuang Jiangu Wan in the treatment of postmenopausal osteoporosis model rats by regulating BMP-2/Smad signaling pathway[J]. Jilin J Tradit Chin Med, 2023, 43(12): 1459-1464. | |
| 2 | 刘爱国, 樊芹英, 郑浩, 等. 藤黄健骨胶囊联合经皮椎体成形术治疗骨质疏松性椎体压缩骨折疗效分析[J]. 中医药临床杂志, 2020, 32(6):1132-1135. |
| LIU A G, FAN Q Y, ZHENG H, et al. Therapeutic effect of Tenghuang Jiangu capsule combined with percutaneous vertebroplasty on osteoporotic vertebral compression fracture[J]. Clin J Tradit Chin Med, 2020, 32(6): 1132-1135. | |
| 3 | 司文腾, 陈宾, 周宇. 藤黄健骨胶囊联合玻璃酸钠治疗膝骨关节炎的临床研究[J]. 现代药物与临床, 2020, 35(3): 525-528. |
| SI W T, CHEN B, ZHOU Y. Clinical study on Tenghuang Jiangu capsules combined with sodium hyaluronate in treatment of knee osteoarthritis[J]. Drugs Clinic, 2020, 35(3): 525-528. | |
| 4 | 杨文博, 乔莉, 刘红喜, 等. 藤黄健骨胶囊在骨质疏松性椎体压缩骨折经皮椎体成形术中的应用[J]. 中华中医药学刊, 2021, 39(10): 215-218. |
| YANG W B, QIAO L, LIU H X, et al. Application of Tenghuang Jiangu capsules combined with PVP for osteoporotic vertebral compression fracture[J]. Chin Archi Tradit Chin Med, 2021, 39(10): 215-218. | |
| 5 | 符仲秋, 王维学, 丰景斌. 藤黄健骨胶囊对大鼠膝骨关节炎的影响[J]. 中国临床药理学杂志, 2021, 37(15): 1996-1998. |
| FU Z Q, WANG W X, FENG J B. Effect of Tenghuang Jiangu capsules on knee osteoarthritis in rats[J]. Chin J Clin Pharmacol, 2021, 37(15): 1996-1998. | |
| 6 | 赵勇, 王艳, 郑金凤, 等. 藤黄健骨丸质量标准研究[J]. 中国药师, 2013, 16(1): 76-80. |
| ZHAO Y, WANG Y, ZHENG J F, et al. Study on the quality standard for Tenghuang Jiangu pills[J]. Chin Pharm, 2013, 16(1): 76-80. | |
| 7 | 赵子剑. HPLC测定藤黄健骨胶囊中淫羊藿苷的含量[J]. 光明中医, 2009, 24(4): 646-647. |
| ZHAO Z J. Determination of icraiin in Tenghuang Jiangu capsules by HPLC[J]. Guangming Tradit Chin Med, 2009, 24(4): 646-647. | |
| 8 | 于曼娜, 张青. 高效液相色谱法测定藤黄健骨胶囊中淫羊藿苷的含量[J]. 海峡药学, 2006, 18(4): 98-100. |
| YU M N, ZHANG Q. Determination of icariin in Tenghuang Jiangu capsules by high performance liquid chromatography[J]. Strait Pharm, 2006, 18(4): 98-100. | |
| 9 | 陈毅鹏, 陈志桃. 高效液相色谱法测定藤黄健骨颗粒中淫羊藿苷的含量[J]. 海峡药学, 2009, 21(10): 47-48. |
| CHEN Y P, CHEN Z T. Determination of icariin in Tenghuang Jiangu granules by HPLC[J]. Strait Pharm, 2009, 21(10): 47-48. | |
| 10 | 于文静, 侯放, 王焕群. 高效液相色谱法测定藤黄健骨丸中淫羊霍苷的含量[J]. 中国药品标准, 2009, 10(3): 210-212. |
| YU W J, HOU F, WANG H Q. Determination of icariin in Tenghuang Jiangu pills by HPLC[J]. Drug Standards China, 2009, 10(3): 210-212. | |
| 11 | 赵昱玮, 南敏伦, 司英奇, 等. 高效液相色谱一测多评法同时检测藤黄健骨丸中7种成分含量[J]. 中国医药导报, 2022, 19(33): 33-36. |
| ZHAO Y W, NAN M L, SI Y Q, et al. Simultaneous determination of 7 components in Tenghuang Jiangu pills by high performance liquid chromatography-quantitative analysis of multi-components by single-marker[J]. Chin Med Her, 2022, 19(33): 33-36. | |
| 12 | 曹辉, 周霖, 孙志, 等. 基于UHPLC-Q-Orbitrap HRMS结合整合网络药理学的藤黄健骨胶囊化学成分识别及作用机制初步研究[J]. 中草药, 2020, 51(9): 2408-2417. |
| CAO H, ZHOU L, SUN Z, et al. Chemical composition identification and mechanism study of Tenghuang Jiangu capsule based on UHPLC-Q-Orbitrap HRMS and network pharmacology[J]. Chin Tradit Herbal Drugs, 2020, 51(9): 2408-2417. | |
| 13 | 李静, 张青, 肖春霞, 等. HPLC波长切换法同时测定排毒养颜胶囊中10种成分[J]. 中草药, 2018, 49(20): 4824-4830. |
| LI J, ZHANG Q, XIAO C X, et al. Simultaneous determination of 10 components in Paidu Yangyan capsules by HPLC wavelength change method[J]. Chin Tradit Herbal Drugs, 2018, 49(20): 4824-4830. | |
| 14 | 陈肖家, 张庆文, 季晖, 等. 紫外分光光度法和高效液相色谱法测定淫羊藿总黄酮含量的比较研究[J]. 药物分析杂志, 2007, 27(5): 625-630. |
| CHEN X J, ZHANG Q W, JI H, et al. Comparison of UV spectrophotometry and HPLC on quantitative determination of total flavonoids of Herba Epimedii[J]. Chin J Pharm Anal, 2007, 27(5): 625-630. | |
| 15 | 丁桂兰, 薛亚光, 邢春来, 等. HPLC测定橘红丸中柚皮苷、橙皮苷的含量[J]. 中成药, 2004, 26(9): 21-24. |
| DING G L, XUE Y G, XING C L, et al. Determination of narigin and hesperidin in Juhong pills by HPLC[J]. Chin Tradit Pat Med, 2004, 26(9): 21-24. | |
| 16 | 周菲, 林美斯, 王琳, 等. 经典名方百合地黄汤物质基准制备及过程质量控制研究[J]. 中草药, 2019, 50(16): 3824-3832. |
| ZHOU F, LIN M S, WANG L, et al. Preparation and process quality control of substance benchmarks of classical famous prescription of Baihe Dihuang Decoction[J]. Chin Tradit Herbal Drugs, 2019, 50(16): 3824-3832. | |
| 17 | 李钦青, 柴金苗, 贺文彬, 等. 高效液相色谱法同时测定复方藿枝颗粒中淫羊藿苷、宝藿苷Ⅰ、甘草苷和甘草酸铵的含量[J]. 中药新药与临床药理, 2020, 31(6): 714-717. |
| LI Q Q, CHAI J M, HE W B, et al. Simultaneous determination of icariin, baohuoside Ⅰ, liquiritin and ammonium glycyrrhizinatein in Fufang Huo Zhi granule by HPLC[J]. Tradit Chin Drug Res Clinic Pharm, 2020, 31(6): 714-717. | |
| 18 | 葛楠, 李志能, 闫广利, 等. 经典名方固阴煎基准样品HPLC指纹图谱研究[J]. 中草药, 2022, 53(24): 7730-7739. |
| GE N, LI Z N, YAN G L, et al. HPLC fingerprinting study of classical formula Guyin Decoction[J]. Chin Tradit Herbal Drugs, 2022, 53(24): 7730-7739. | |
| 19 | 孙庆妹, 王琼晓, 尤金玲, 等. 基于HPLC指纹图谱和多成分定量的玄参质量评价研究[J]. 中草药, 2023, 54(20): 6827-6835. |
| SUN Q M, WANG Q X, YOU J L, et al. Quality evaluation of Scrophularia ningpoensis based on HPLC fingerprint and multi-component content determination[J]. Chin Tradit Herbal Drugs, 2023, 54(20): 6827-6835. |
| [1] | Ye-Chen WANG, Zhi-Dong QIU, Yan-Xu ZHANG, Li-Ying WANG, Mao-Xu WANG, Xue-Lian DONG. Research on Quality Evaluation of Yiyi-Zhuye Powders Based on Fingerprint, Quality Value Transfer and Chemometrics [J]. Chinese Journal of Applied Chemistry, 2024, 41(3): 422-436. |
| [2] | Zhong-Yi LIU, Cong LIU, Yong-Fan MA, Wei ZHAO, Yan-Hua LIU, Hong-Bo CHU. Fingerprints and Action Mechanism of Efficacy-Associated Substances of Yishen Paishi Formula [J]. Chinese Journal of Applied Chemistry, 2023, 40(11): 1558-1571. |
| [3] | LI Wenjie, WANG Lixue, SUN Linghui, YOU Wei, ZHAO Yabin. Progress in Matrix-Assisted Laser Desorption Mass Spectrometry Imaging for Fingerprint Analysis [J]. Chinese Journal of Applied Chemistry, 2020, 37(10): 1137-1146. |
| [4] | JIA Zehui1, WANG Chuntao1, LI Hua2*. The Applications of Multivariate Curve Resolution-alternating Least Squares in Resolving Overlapped Spectra of Corydalis yanhusuo Fingerprint and the Content Detection of Tetrahydropalmatine [J]. Chinese Journal of Applied Chemistry, 2013, 30(03): 329-334. |
| [5] | YU Xuejiao, LIU Jianjun*, YU Yingchun, ZUO Shengli. Application of Fluorescent Quantum Dots Synthesized via Chemical Routes in Fingerprints Development [J]. Chinese Journal of Applied Chemistry, 2012, 29(08): 855-862. |
| [6] | ZHANG Meiqin*, ZHANG Ting, QIN Gang, ZHANG Yang, ZHANG Xueji. Progress in the detection methods of blood fingerprints [J]. Chinese Journal of Applied Chemistry, 2012, 29(01): 1-8. |
| [7] | CHEN Yan1,2, ZHANG Chunjing2, GAO Dongmei2, YANG Fan2, HAN Dongxue2* , NIU Li2*. Development of Visualization of Latent Fingerprints [J]. Chinese Journal of Applied Chemistry, 2011, 28(10): 1099-1107. |
| [8] | XIA Binbin1, YANG Ruiqin1*, WANG Yanji1, LIU Jianjun2. CdSe Quantum Dots Labeling to Blood Fingerprints on Dark Surfaces and Fluorescent Imaging [J]. Chinese Journal of Applied Chemistry, 2011, 28(06): 689-694. |
| [9] | LIANG Hua-Zheng1, YANG Shui-Ping1, CHEN Shuang-Xi1, CHEN Huan-Wen1,2*, LIU Qing3. Rapidly Fingerprinting Condiment Vegetables Using Surface Desorption Atomospheric Pressure Chemical Ionization Mass Spectrometry [J]. Chinese Journal of Applied Chemistry, 2010, 27(05): 606-610. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||