应用化学 ›› 2023, Vol. 40 ›› Issue (3): 413-419.DOI: 10.19894/j.issn.1000-0518.220212
甲醛次硫酸氢钠二水合物促进的醌芳基化反应
- 1.江苏警官学院安检排爆实验室,南京 210031
2.常州工程职业技术学院化工与制药工程学院,常州 213164
3.山东天宝化工股份有限公司,平邑 273300
Rongalite Promoted Arylation of Benzoquinone
Min-Xiao XU1, Liang WANG2( ), Cheng-Fang CHEN3
), Cheng-Fang CHEN3
- 1.Security Clearance Lab,Jiangsu Police Institute,Nanjing 210031,China
2.School of Chemical and Pharmaceutical Engineering,Changzhou Vocational Institute of Engineering,Changzhou 213164,China
3.Shandong Tian Bao Chemical Co. LTD,Pingyi 273300,China
摘要:
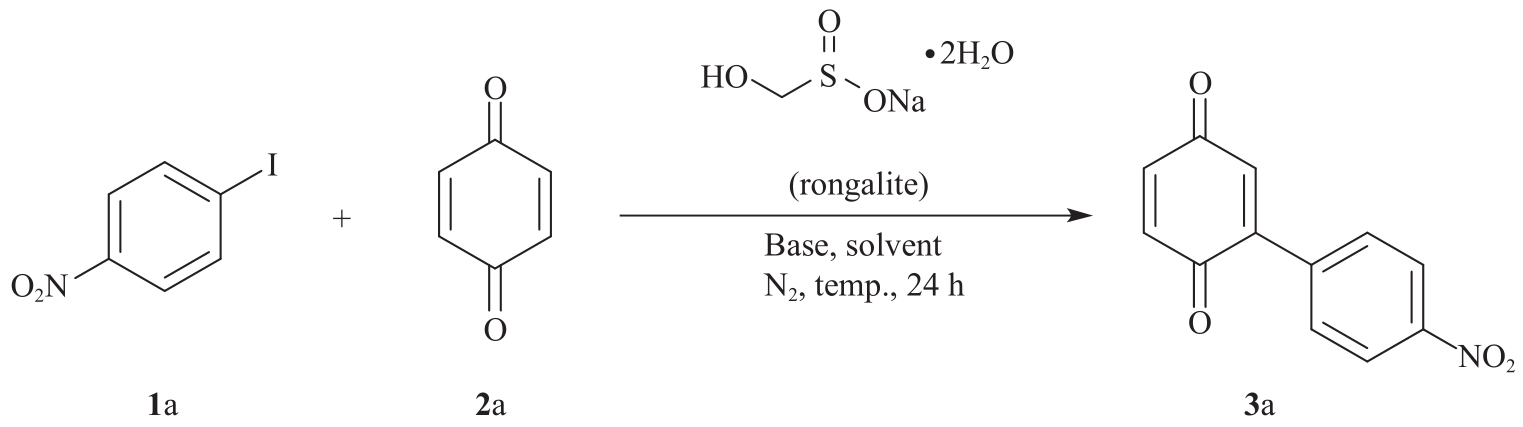
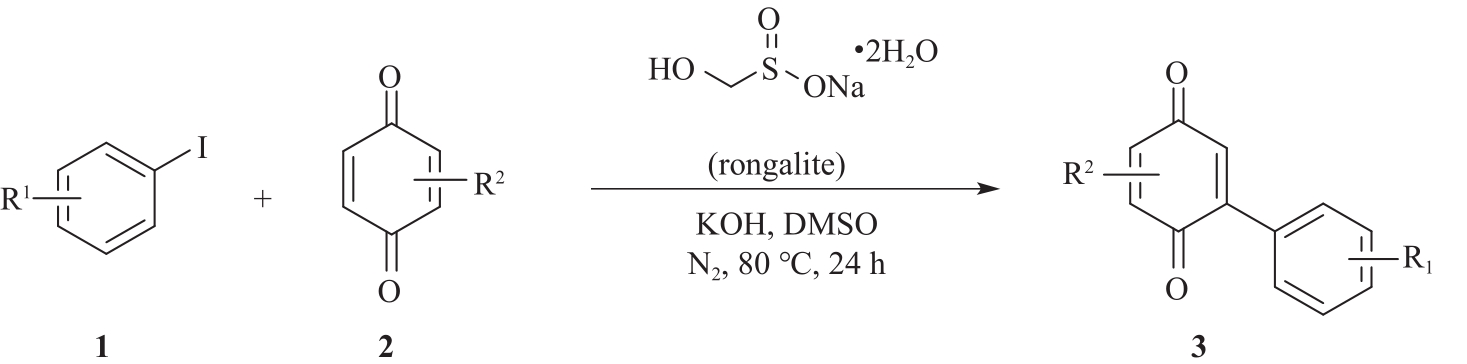
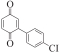
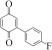
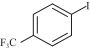
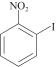
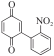
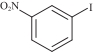
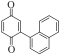
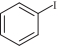
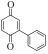
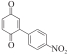
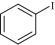
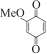
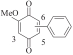
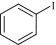
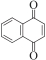
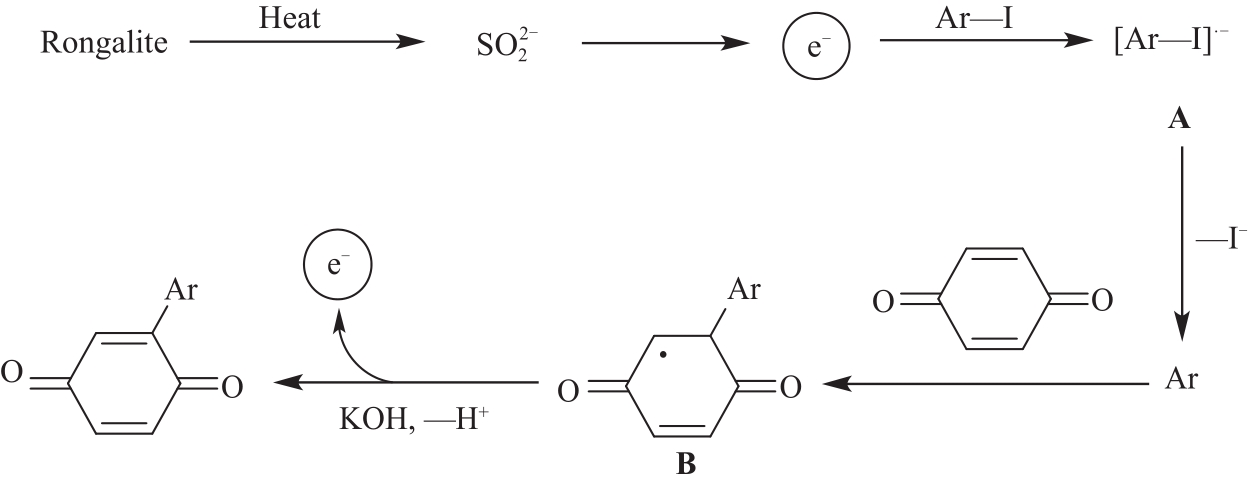
以甲醛次硫酸氢钠二水合物(rongalite)为电子供体,碘代芳烃为芳基化试剂,在温和条件下实现了醌的芳基化反应。以苯醌和对硝基碘苯的反应为模板反应,考察了碱、溶剂、温度和物料比等对反应的影响。结果表明,最佳合成条件为:对硝基碘苯(0.2 mmol),苯醌(1 mmol),甲醛次硫酸氢钠二水合物(0.4 mmol),氢氧化钾(0.6 mmol),DMSO(1 mL),氮气氛围,80 ℃,反应24 h。在该条件下,目标产物收率可达82%。该反应体系对不同的碘代芳烃和醌均表现出了良好的适用性,收率在62%~84%之间。
中图分类号: